NTA JEE Main 5th September 2020 Shift 1
For the following questions answer them individually
NTA JEE Main 5th September 2020 Shift 1 - Question 31
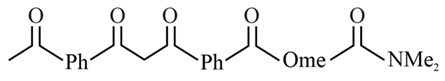
The increasing order of the acidity of the $$\alpha$$-hydrogen of the following compounds is:
NTA JEE Main 5th September 2020 Shift 1 - Question 32
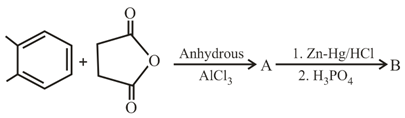
In the following reaction sequence the major products A and B are:
NTA JEE Main 5th September 2020 Shift 1 - Question 33
The condition that indicates a polluted environment is:
NTA JEE Main 5th September 2020 Shift 1 - Question 34
A diatomic molecule $$\text{X}_2$$ has a body-centred cubic (bcc) structure with a cell edge of $$300\,\text{pm}$$. The density of the molecule is $$6.17\,\text{g cm}^{-3}$$. The number of molecules present in $$200\,\text{g}$$ of $$\text{X}_2$$ is: (Avogadro constant $$(N_A) = 6 \times 10^{23}\,\text{mol}^{-1}$$)
NTA JEE Main 5th September 2020 Shift 1 - Question 35
A flask contains a mixture of compounds A and B. Both compounds decompose by first-order kinetics. The half-lives for A and B are $$300\,\text{s}$$ and $$180\,\text{s}$$, respectively. If the concentrations of A and B are equal initially, the time required for the concentration of A to be four times that of B (in s) is: (Use $$\ln 2 = 0.693$$)
NTA JEE Main 5th September 2020 Shift 1 - Question 36
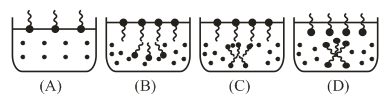
Identify the correct molecular picture showing what happens at the critical micellar concentration (CMC) of an aqueous solution of a surfactant.
NTA JEE Main 5th September 2020 Shift 1 - Question 37
An Ellingham diagram provides information about:
NTA JEE Main 5th September 2020 Shift 1 - Question 38
The structure of $$\text{PCl}_5$$ in the solid state is:
NTA JEE Main 5th September 2020 Shift 1 - Question 39
The correct electronic configuration and spin-only magnetic moment (BM) of $$\text{Gd}^{3+}$$ ($$Z = 64$$), respectively, are:
NTA JEE Main 5th September 2020 Shift 1 - Question 40
The values of the crystal field stabilization energies for a high spin $$d^6$$ metal ion in octahedral and tetrahedral fields, respectively, are:


