NTA JEE Main 4th September 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 26
The shortest wavelength of H atom in the Lyman series is $$\lambda_1$$. The longest wavelength in the Balmer series of He$$^+$$ is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 27
The process that is NOT endothermic in nature is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 28
The reaction in which the hybridisation of the underlined atom is affected is
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 29
The molecule in which hybrid MOs involve only one d-orbital of the central atom is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 30
Five moles of an ideal gas at 1 bar and 298 K is expanded into vacuum to double the volume. The work done is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 31
If the equilibrium constant for $$A \rightleftharpoons B + C$$ is $$K_{eq}^{(1)}$$ and that of $$B + C \rightleftharpoons P$$ is $$K_{eq}^{(2)}$$, the equilibrium constant for $$A \rightleftharpoons P$$ is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 32
An alkaline earth metal 'M' readily forms water soluble sulphate and water insoluble hydroxide. Its oxide MO is very stable to heat and does not have rock-salt structure. M is
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 33
Which of the following compounds will form the precipitate with aq. AgNO$$_3$$ solution most readily?
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 34
Among the following compounds, which one has the shortest C-Cl bond?
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 35
The processes of calcination and roasting in metallurgical industries, respectively, can lead to:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 36
250 mL of a waste solution obtained from the workshop of a goldsmith contains 0.1 M AgNO$$_3$$ and 0.1 M AuCl. The solution was electrolyzed at 2 V by passing a current of 1 A for 15 minutes. The metal/metals electrodeposited will be: $$\left(E^0_{Ag^+/Ag} = 0.80V, E^0_{Au^+/Au} = 1.69V\right)$$
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 37
A sample of red ink (a colloidal suspension) is prepared by mixing eosine dye, egg white, HCHO and water. The component which ensures stability of the ink sample is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 38
The incorrect statement(s) among (a) - (c) is (are):
(a) W(VI) is more stable than Cr(VI).
(b) in the presence of HCl, permanganate titrations provide satisfactory results.
(c) some lanthanoid oxides can be used as phosphors.
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 39
The Crystal Field Stabilization Energy (CFSE) of $$[CoF_3(H_2O)_3]$$ $$(\Delta_0 < P)$$ is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 40
The one that can exhibit highest paramagnetic behaviour among the following is: gly = glycinato; bpy = 2, 2'-bipyridine
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 41
The major product [B] in the following reaction is:
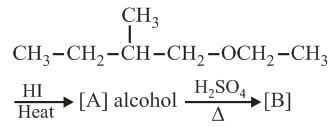
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 42
The major product [R] in the following sequence of reaction is:
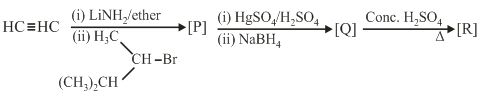
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 43
The major product [C] of the following reaction sequence will be:
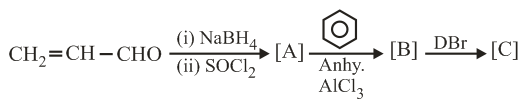
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 44
In the following reaction sequence, [C] is:
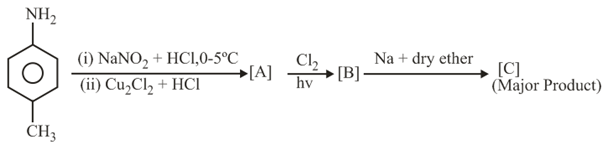
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 45
The mechanism of action of "Terfenadine" (Seldane) is:
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 46
A 100 mL solution was made by adding 1.43 g of $$Na_2CO_3 \cdot xH_2O$$. The normality of the solution is 0.1 N. The value of x is __________ (The atomic mass of Na is 23 g/mol)
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 47
Consider the following equations:
$$2Fe^{2+} + H_2O_2 \to xA + yB$$ (in basic medium)
$$2MnO_4^- + 6H^+ + 5H_2O_2 \to x'C + y'D + z'E$$ (in acidic medium)
The sum of the stoichiometric coefficients x, y, x', y' and z' for products A, B, C, D and E respectively, is
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 48
The osmotic pressure of a solution of NaCl is 0.10 atm and that of a glucose solution is 0.20 atm. The osmotic pressure of a solution formed by mixing 1 L of the sodium chloride solution with 2 L of the glucose solution is $$x \times 10^{-3}$$ atm. x is __________ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 49
The number of molecules with energy greater than the threshold energy for a reaction increases five fold by a rise of temperature from 27°C to 42°C. Its energy of activation in J/mol is __________ (Take ln 5 = 1.6094; R = 8.314 J mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 2 - Chemistry - Question 50
The number of chiral centres present in threonine is __________
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




