NTA JEE Main 4th September 2020 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 26
The region in the electromagnetic spectrum where the Balmer series lines appear is:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 27
The elements with atomic numbers 101 and 104 belong to, respectively:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 28
The ionic radii of $$O^{2-}$$, $$F^-$$, $$Na^+$$ and $$Mg^{2+}$$ are in the order:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 29
The intermolecular potential energy for the molecules A, B, C and D given below suggests that:
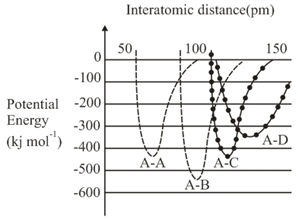
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 30
For one mole of an ideal gas, which of these statements must be true?
(a) Internal energy (U) and enthalpy (H) each depends on temperature.
(b) Compressibility factor Z is not equal to 1
(c) $$C_{P,m} - C_{V,m} = R$$
(d) $$dU = C_v dT$$ for any process
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 31
On combustion of Li, Na and K in excess of air, the major oxides formed, respectively, are:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 32
On heating, lead (II) nitrate gives a brown gas (A). The gas (A) on cooling changes to a colourless solid/liquid (B). (B) on heating with NO changes to a blue solid (C). The oxidation number of nitrogen in solid (C) is:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 33
The IUPAC name of the following compound is:
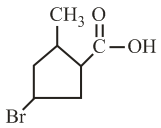
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 34
The decreasing order of reactivity of the following organic molecules towards AgNO$$_3$$ solution is:
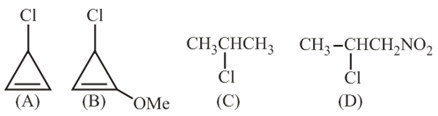
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 35
Identify the incorrect statement from the options below for the above cell:
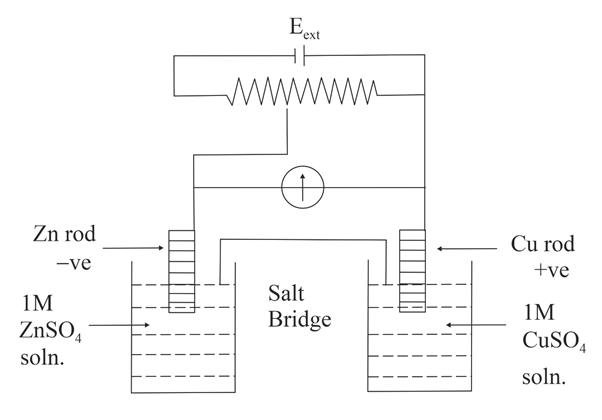
$$E^0_{Cu^{2+}|Cu} = +0.34$$ V,
$$E^0_{Zn^{2+}|Zn} = -0.76$$ V
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 36
For the equilibrium $$A \rightleftharpoons B$$, the variation of the rate of the forward (a) and reverse (b) reaction with time is given by:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 37
Match the following:
(i) Foam (a) smoke
(ii) Gel (b) cell fluid
(iii) Aerosol (c) jellies
(iv) Emulsion (d) rubber
(e) froth
(f) milk
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 38
Among statements (a) - (d), the correct ones are:
(a) Lime stone is decomposed to CaO during the extraction of iron from its oxides.
(b) In the extraction of silver, silver is extracted as an anionic complex.
(c) Nickel is purified by Mond's process.
(d) Zr and Ti are purified by Van Arkel method.
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 39
The number of isomers possible for $$[Pt(en)(NO_2)_2]$$ is:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 40
The pair in which both the species have the same magnetic moment (spin only) is:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 41
An organic compound (A) (molecular formula $$C_6H_{12}O_2$$) was hydrolysed with dil. $$H_2SO_4$$ to give a carboxylic acid (B) and an alcohol (C). 'C' gives white turbidity immediately when treated with anhydrous $$ZnCl_2$$ and conc. HCl. The organic compound (A) is:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 42
When neopentyl alcohol is heated with an acid, it slowly converted into an 85:15 mixture of alkenes A and B, respectively. What are these alkenes?
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 43
[P] on treatment with $$Br_2/FeBr_3$$ in $$CCl_4$$ produced a single isomer $$C_8H_7O_2Br$$ while heating [P] with sodalime gives toluene. The compound [P] is:
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 44
Which of the following will react with $$CHCl_3$$ + alc. KOH?
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 45
What are the functional groups present in the structure of maltose?
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 46
The mass of ammonia in grams produced when 2.8 kg of dinitrogen quantitatively reacts with 1 kg of dihydrogen is __________
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 47
A 20.0 mL solution containing 0.2 g impure $$H_2O_2$$ reacts completely with 0.316 g of $$KMnO_4$$ in acid solution. The purity of $$H_2O_2$$ (in %) is __________ (mol. wt. of $$H_2O_2$$ = 34; mol. wt. of $$KMnO_4$$ = 158)
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 48
At 300 K, the vapour pressure of a solution containing 1 mole of n-hexane and 3 moles of n-heptane is 550 mm of Hg. At the same temperature, if one more mole of n-heptane is added to this solution, the vapour pressure of the solution increases by 10 mm of Hg. What is the vapour pressure in mmHg of n-heptane in its pure state?
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 49
If 75% of a first order reaction was completed in 90 minutes, 60% of the same reaction would be completed in approximately (in minutes) __________ (Take: log 2 = 0.30; log 2.5 = 0.40)
789
456
123
0.-
Clear All
NTA JEE Main 4th September 2020 Shift 1 - Chemistry - Question 50
The number of chiral centres present in [B] is __________
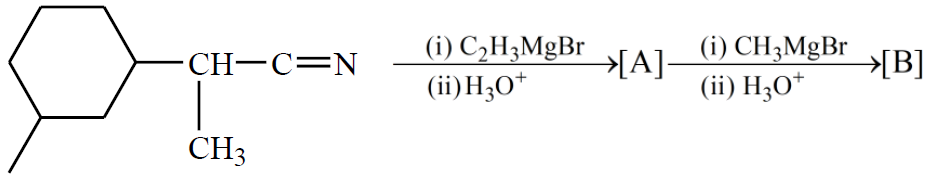
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




