NTA JEE Main 31st January 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 31st January 2023 Shift 1 - Question 51
On complete combustion, 0.492 g of an organic compound gave 0.792 g of CO$$_2$$. The % of carbon in the organic compound is (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 52
Zinc reacts with hydrochloric acid to give hydrogen and zinc chloride. The volume of hydrogen gas produced at STP from the reaction of 11.5 g of zinc with excess HCl is L (Nearest integer)
(Given: Molar mass of Zn is 65.4 g mol$$^{-1}$$ and Molar volume of H$$_2$$ at STP = 22.7 L)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 53
The enthalpy change for the conversion of $$\dfrac{1}{2}$$Cl$$_2$$(g) to Cl$$^-$$(aq) is (-) ______ kJmol$$^{-1}$$ (Nearest integer)
Given: $$\Delta_{dis}H^0_{Cl_2(g)} = 240$$ kJmol$$^{-1}$$
$$\Delta_{eg}H^o_{Cl(g)} = -350$$ kJmol$$^{-1}$$
$$\Delta_{hyd}H^o_{Cl^-(g)} = -380$$ kJmol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 54
For reaction: $$SO_2(g) + \dfrac{1}{2}O_2(g) \rightleftharpoons SO_3(g)$$ $$K_P = 2 \times 10^{12}$$ at 27°C and 1 atm pressure. The $$K_c$$ for the same reaction is ______ $$\times 10^{13}$$. (Nearest integer)
(Given $$R = 0.082$$ L atm K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 55
The total pressure of a mixture of non-reacting gases X(0.6 g) and Y(0.45 g) in a vessel is 740 mm of Hg. The partial pressure of the gas X is mm of Hg. (Nearest Integer)
(Given: molar mass X = 20 and Y = 45 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 56
At 27°C, a solution containing 2.5 g of solute in 250.0 mL of solution exerts an osmotic pressure of 400 Pa. The molar mass of the solute is g mol$$^{-1}$$ (Nearest integer)
(Given: R = 0.083 L bar$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 57
The logarithm of equilibrium constant for the reaction $$Pd^{2+} + 4Cl^- \rightleftharpoons PdCl_4^{2-}$$ is (Nearest integer)
Given: $$\dfrac{2.303RT}{F} = 0.06$$ V
$$Pd^{2+}_{(aq)} + 2e^- \rightleftharpoons Pd(s)$$ $$E^o = 0.83$$ V
$$PdCl_4^{2-}(aq) + 2e^- \rightleftharpoons Pd(s) + 4Cl^-(aq)$$ $$
$$E^o = 0.65$$ V
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 58
$$A \rightarrow B$$
The rate constants of the above reaction at 200 K and 300 K are 0.03 min$$^{-1}$$ and 0.05 min$$^{-1}$$ respectively. The activation energy for the reaction is J (Nearest integer)
(Given: ln 10 = 2.3, R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$, log 5 = 0.70, log 3 = 0.48, log 2 = 0.30)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Question 59
The oxidation state of phosphorus in hypophosphoric acid is
789
456
123
0.-
Clear All
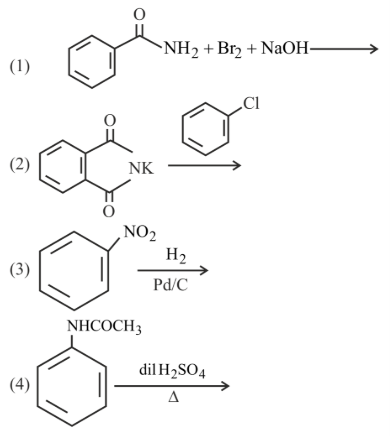
NTA JEE Main 31st January 2023 Shift 1 - Question 60
How many of the transformation given below would result in aromatic amines?
789
456
123
0.-
Clear All


