NTA JEE Main 31st January 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 31
Which transition in the hydrogen spectrum would have the same wavelength as the Balmer type transition from n = 4 to n = 2 of He$$^+$$ spectrum
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 32
The correct increasing order of the ionic radii is
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 33
Match List I with List II
List I List II
A. XeF$$_4$$ I. See-saw
B. SF$$_4$$ II. Square planar
C. NH$$_4^+$$ III. Bent T-shaped
D. BrF$$_3$$ IV. Tetrahedral
Choose the correct answer from the options given below:
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 34
$$H_2O_2$$ acts as a reducing agent in
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 35
Match items of column I and II
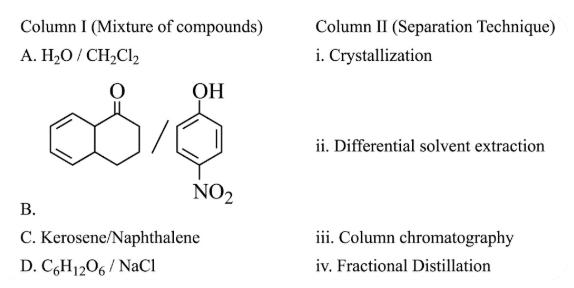
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 36
Choose the correct set of reagents for the following conversion
trans Ph - CH = CH - CH$$_3$$ $$\rightarrow$$ cis Ph - CH = CH - CH$$_3$$
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 37
Which one of the following statements is correct for electrolysis of brine solution?
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 38
Adding surfactants in non polar solvent, the micelles structure will look like
(a)
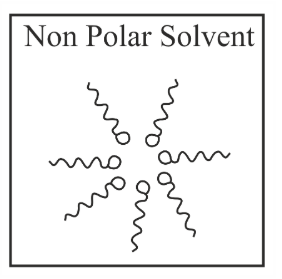
(b)
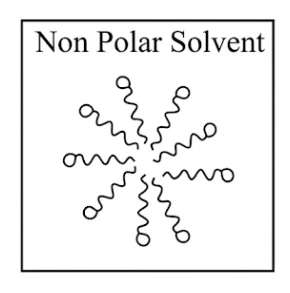
(c)
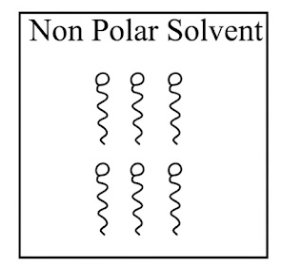
(d)
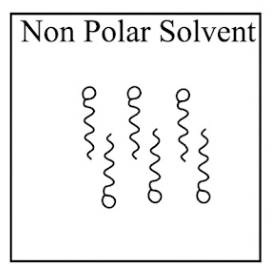
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 39
The methods NOT involved in concentration of ore are
(A) Liquation
(B) Leaching
(C) Electrolysis
(D) Hydraulic washing
(E) Froth floatation
Choose the correct answer from the options given below:
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 40
Identify X, Y and Z in the following reaction. (Equation not balanced)
$$ClO^. + NO_2 \rightarrow X \xrightarrow{H_2O} Y + Z$$
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 41
When Cu$$^{2+}$$ ion is treated with KI, a white precipitate, X appears in solution. The solution is titrated with sodium thiosulphate, the compound Y is formed. X and Y respectively are
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 42
The correct order of basicity of oxides of vanadium is
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 43
Nd$$^{2+}$$ = ______
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 44
Cobalt chloride when dissolved in water forms pink colored complex X which has octahedral geometry. This solution on treating with conc HCl forms deep blue complex, Y which has a Z geometry. X, Y and Z, respectively, are
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 45
The correct order of melting point of dichlorobenzenes is
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 46
An organic compound 'A' with empirical formula C$$_6$$H$$_6$$O gives sooty flame on burning. Its reaction with bromine solution in low polarity solvent results in high yield of B. B is
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 47
Consider the following reaction
Propanal + Methanal $$\xrightarrow{(i)$$ dil. NaOH $$}{\xrightarrow{(ii) \Delta \; (iii)$$ NaCN $$\; (iv) \; H_3O^+}}$$ Product B (C$$_5$$H$$_8$$O$$_3$$)
The correct statement for product B is. It is
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 48
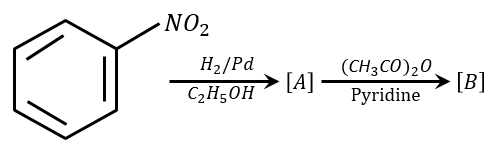
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 49
Which of the following artificial sweeteners has the highest sweetness value in comparison to cane sugar?
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 50
A protein 'X' with molecular weight of 70,000 u, on hydrolysis gives amino acids. One of these amino acid is
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 51
On complete combustion, 0.492 g of an organic compound gave 0.792 g of CO$$_2$$. The % of carbon in the organic compound is (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 52
Zinc reacts with hydrochloric acid to give hydrogen and zinc chloride. The volume of hydrogen gas produced at STP from the reaction of 11.5 g of zinc with excess HCl is L (Nearest integer)
(Given: Molar mass of Zn is 65.4 g mol$$^{-1}$$ and Molar volume of H$$_2$$ at STP = 22.7 L)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 53
The enthalpy change for the conversion of $$\dfrac{1}{2}$$Cl$$_2$$(g) to Cl$$^-$$(aq) is (-) ______ kJmol$$^{-1}$$ (Nearest integer)
Given: $$\Delta_{dis}H^0_{Cl_2(g)} = 240$$ kJmol$$^{-1}$$
$$\Delta_{eg}H^o_{Cl(g)} = -350$$ kJmol$$^{-1}$$
$$\Delta_{hyd}H^o_{Cl^-(g)} = -380$$ kJmol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 54
For reaction: $$SO_2(g) + \dfrac{1}{2}O_2(g) \rightleftharpoons SO_3(g)$$ $$K_P = 2 \times 10^{12}$$ at 27°C and 1 atm pressure. The $$K_c$$ for the same reaction is ______ $$\times 10^{13}$$. (Nearest integer)
(Given $$R = 0.082$$ L atm K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 55
The total pressure of a mixture of non-reacting gases X(0.6 g) and Y(0.45 g) in a vessel is 740 mm of Hg. The partial pressure of the gas X is mm of Hg. (Nearest Integer)
(Given: molar mass X = 20 and Y = 45 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 56
At 27°C, a solution containing 2.5 g of solute in 250.0 mL of solution exerts an osmotic pressure of 400 Pa. The molar mass of the solute is g mol$$^{-1}$$ (Nearest integer)
(Given: R = 0.083 L bar$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 57
The logarithm of equilibrium constant for the reaction $$Pd^{2+} + 4Cl^- \rightleftharpoons PdCl_4^{2-}$$ is (Nearest integer)
Given: $$\dfrac{2.303RT}{F} = 0.06$$ V
$$Pd^{2+}_{(aq)} + 2e^- \rightleftharpoons Pd(s)$$ $$E^o = 0.83$$ V
$$PdCl_4^{2-}(aq) + 2e^- \rightleftharpoons Pd(s) + 4Cl^-(aq)$$ $$
$$E^o = 0.65$$ V
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 58
$$A \rightarrow B$$
The rate constants of the above reaction at 200 K and 300 K are 0.03 min$$^{-1}$$ and 0.05 min$$^{-1}$$ respectively. The activation energy for the reaction is J (Nearest integer)
(Given: ln 10 = 2.3, R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$, log 5 = 0.70, log 3 = 0.48, log 2 = 0.30)
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 59
The oxidation state of phosphorus in hypophosphoric acid is
789
456
123
0.-
Clear All
NTA JEE Main 31st January 2023 Shift 1 - Chemistry - Question 60
How many of the transformation given below would result in aromatic amines?
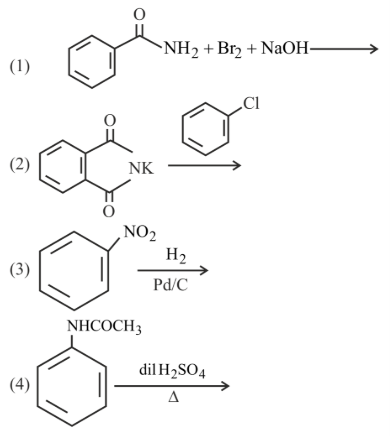
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




