NTA JEE Main 30th January 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 31
Match List - I with List - II
| LIST-I (Atomic number) | LIST-II (Block of periodic table) |
|---|---|
| (A) 37 | I. p-block |
| (B) 78 | II. d-block |
| (C) 52 | III. f-block |
| (D) 65 | IV. s-block |
Choose the correct answer from the options given below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 32
For OF$$_2$$ molecule consider the following:
(A) Number of lone pairs on oxygen is 2.
(B) FOF angle is less than 104.5°.
(C) Oxidation state of O is -2.
(D) Molecule is bent 'V' shaped.
(E) Molecular geometry is linear.
Correct options are:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 33
Match List I with List II
| List I (molecules/ions) | List II (No. of lone pairs of e$$^-$$ on central atom) |
|---|---|
| (A) IF$$_7$$ | I. Three |
| (B) ICl$$_4^-$$ | II. One |
| (C) XeF$$_6$$ | III. Two |
| (D) XeF$$_2$$ | IV. Zero |
Choose the correct answer from the options given below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 34
The alkaline earth metal sulphate(s) which are readily soluble in water is/are:
(A) BeSO$$_4$$
(B) MgSO$$_4$$
(C) CaSO$$_4$$
(D) SrSO$$_4$$
(E) BaSO$$_4$$
Choose the correct answer from the options below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 35
Lithium aluminium hydride can be prepared from the reaction of
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 36
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): In expensive scientific instruments, silica gel is kept in watch-glasses or in semipermeable membrane bags.
Reason (R): Silica gel adsorbs moisture from air via adsorption, thus protects the instrument from water corrosion (rusting) and / or prevents malfunctioning.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 37
What is the correct order of acidity of the protons marked $$A - D$$ in the given compounds?
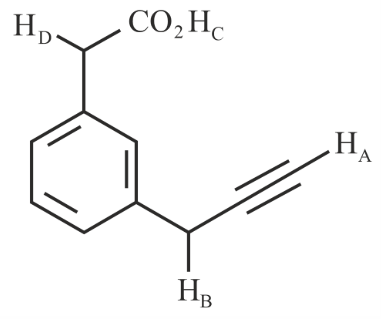
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 38
The major products 'A' and 'B', respectively, are
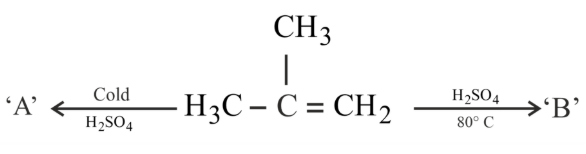
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 39
Formation of photochemical smog involves the following reaction in which A, B and C are respectively.
(i) NO$$_2$$ $$\xrightarrow{h\nu}$$ A + B
(ii) B + O$$_2$$ $$\to$$ C
(iii) A + C $$\to$$ NO$$_2$$ + O$$_2$$
Choose the correct answer from the options below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 40
In the extraction of copper, its sulphide ore is heated in a reverberatory furnace after mixing with silica to:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 41
During the qualitative analysis of SO$$_3^{2-}$$ using dilute H$$_2$$SO$$_4$$, SO$$_2$$ gas is evolved which turns K$$_2$$Cr$$_2$$O$$_7$$ solution (acidified with dilute H$$_2$$SO$$_4$$):
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 42
Which of the following is correct order of ligand field strength?
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 43
To inhibit the growth of tumours, identify the compounds used from the following:
(A) EDTA
(B) Coordination Compounds of Pt
(C) D-Penicillamine
(D) Cis - Platin
Choose the correct answer from the option given below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 44
Match List I with List II
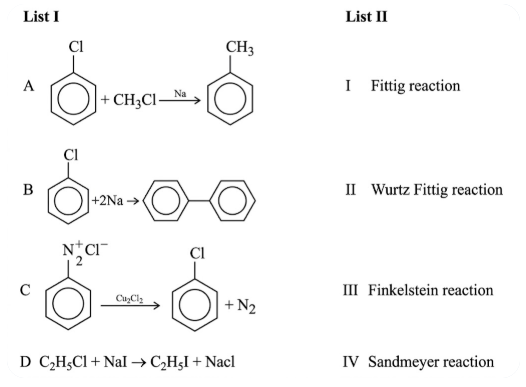
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 45
Which of the following compounds would give the following set of qualitative analysis?
(i) Fehling's Test: Positive
(ii) Na fusion extract upon treatment with sodium nitroprusside gives a blood red colour but not prussian blue.
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 46
Benzyl isocyanide can be obtained by:
(A)
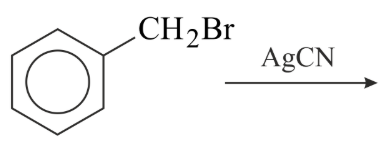
(B)
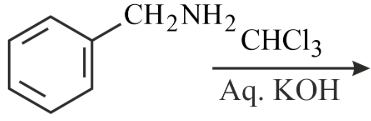
(C)
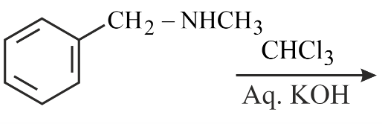
(D)
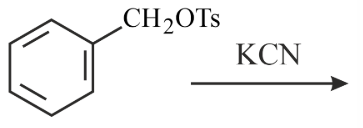
Choose the correct answer from the options given below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 47
Caprolactam when heated at high temperature in presence of water, gives
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 48
Amongst the following compounds, which one is an antacid?
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 49
In the wet tests for identification of various cations by precipitation, which transition element cation doesn't belong to group IV in qualitative inorganic analysis?
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 50
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Ketoses give Seliwanoff's test faster than Aldoses.
Reason (R): Ketoses undergo $$\beta$$-elimination followed by formation of furfural.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 51
The energy of one mole of photons of radiation of frequency $$2 \times 10^{12}$$ Hz in J mol$$^{-1}$$ is ______ . (Nearest integer)
(Given: h $$= 6.626 \times 10^{-34}$$ Js, N$$_A = 6.022 \times 10^{23}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 52
When 2 litre of ideal gas expands isothermally into vacuum to a total volume of 6 litre, the change in internal energy is ______ J. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 53
600 mL of 0.01 M HCl is mixed with 400 mL of 0.01 M H$$_2$$SO$$_4$$. The pH of the mixture is ______ $$\times 10^{-2}$$. (Nearest integer)
[Given log2 = 0.30, log3 = 0.48, log5 = 0.69, log7 = 0.84, log11 = 1.04]
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 54
A 300 mL bottle of soft drink has 0.2M CO$$_2$$ dissolved in it. Assuming CO$$_2$$ behaves as an ideal gas, the volume of the dissolved CO$$_2$$ at STP is ______ mL. (Nearest integer)
Given: At STP, molar volume of an ideal gas is 22.7 L mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 55
A solution containing 2 g of a non-volatile solute in 20 g of water boils at 373.52 K. The molecular mass of the solute is ______ g mol$$^{-1}$$. (Nearest integer)
Given, water boils at 373 K, K$$_b$$ for water $$= 0.52$$ K kg mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 56
Some amount of dichloromethane CH$$_2$$Cl$$_2$$ is added to 671.141 mL of chloroform CHCl$$_3$$ to prepare $$2.6 \times 10^{-3}$$ M solution of CH$$_2$$Cl$$_2$$ DCM. The concentration of DCM is ______ ppm (by mass).
Given: Atomic mass: C = 12; H: 1; Cl = 35.5 density of CHCl$$_3$$ = 1.49 g cm$$^{-3}$$
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 57
Consider the cell
Pt, H$$_2$$(g, 1 atm) | H$$^+$$ aq, 1M | Fe$$^{3+}$$ aq, Fe$$^{2+}$$ aq | Pt s
When the potential of the cell is 0.712 V at 298 K, the ratio Fe$$^{2+}$$/Fe$$^{3+}$$ is ______ (Nearest integer)
Given: Fe$$^{3+}$$ + e$$^-$$ = Fe$$^{2+}$$, E° Fe$$^{3+}$$, Fe$$^{2+}$$ Pt = 0.771, $$\frac{2.303RT}{F} = 0.06$$ V
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 58
If compound A reacts with B following first order kinetics with rate constant $$2.011 \times 10^{-3}$$ s$$^{-1}$$. The time taken by A (in seconds) to reduce from $$7$$ g to $$2$$ g will be ______. (Nearest Integer)
log5 = 0.698, log7 = 0.845, log2 = 0.301
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 59
The number of electrons involved in the reduction of permanganate to manganese dioxide in acidic medium is ______.
789
456
123
0.-
Clear All
NTA JEE Main 30th January 2023 Shift 1 - Chemistry - Question 60
A trisubstituted compound 'A', C$$_{10}$$H$$_{12}$$O$$_2$$ gives neutral FeCl$$_3$$ test positive. Treatment of compound 'A' with NaOH and CH$$_3$$Br gives C$$_{11}$$H$$_{14}$$O$$_2$$, with hydroiodic acid gives methyl iodide and with hot conc. NaOH gives a compound B, C$$_{10}$$H$$_{12}$$O$$_2$$. Compound 'A' also decolourises alkaline KMnO$$_4$$. The number of $$\pi$$ bond/s present in the compound 'A' is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




