NTA JEE Main 3rd September 2020 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 26
The strengths of 5.6 volume hydrogen peroxide (of density 1 g/mL) in terms of mass percentage and molarity(M) respectively, are: (Take molar mass of hydrogen peroxide as 34 g/mol)
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 27
Consider the hypothetical situation where the azimuthal quantum number, $$\ell$$, takes values 0, 1, 2, ......... n + 1. Where n is the principal quantum number. Then, the element with atomic number:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 28
The five successive ionization enthalpies of an element are 800, 2427, 3658, 35024, 32824 kJ mol$$^{-1}$$. The number of valence electrons in the element is:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 29
Consider the following molecules and statements related to them:
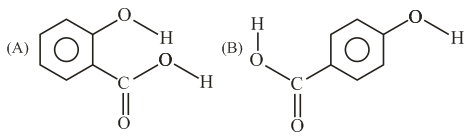
(a) (B) is more likely to be crystalline than (A)
(b) (B) has higher boiling point than (A)
(c) (B) dissolves more readily than (A) in water
Identify the correct option from below:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 30
A mixture of one mole each of $$H_2$$, He and $$O_2$$ each are enclosed in a cylinder of volume V at temperature T. If the partial pressure of $$H_2$$ is 2 atm, the total pressure of the gases in the cylinder is:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 31
100 mL of 0.1 M HCl is taken in a beaker and to it 100 mL 0.1 M NaOH of is added in steps of 2 mL and the pH is continuously measured. Which of the following graphs correctly depicts the change in pH?
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 32
Among the statements (I - IV), the correct ones are:
(I) Be has smaller atomic radius compared to Mg.
(II) Be has higher ionization enthalpy than Al.
(III) Charge/radius ratio of Be is greater than that of Al.
(IV) Both Be and Al form mainly covalent compounds
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 33
The incorrect statement(s) among (a) - (d) regarding acid rain is (are):
(a) It can corrode water pipes
(b) It can damage structures made up of stone.
(c) It cannot cause respiratory ailments in animals
(d) It is not harmful for trees
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 34
For the reaction $$2A + 3B + \frac{3}{2}C \to 3P$$, which statement is correct?
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 35
The incorrect statement is:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 36
Complex A has a composition of $$H_{12}O_6Cl_3Cr$$. If the complex on treatment with conc. $$H_2SO_4$$ loses 13.5% of its original mass, the correct molecular formula of A is:
[Given: atomic mass of Cr = 52 amu and Cl = 35 amu]
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 37
The d-electron configuration of $$[Ru(en)_3]Cl_2$$, and $$[Fe(H_2O)_6]Cl_2$$ respectively are:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 38
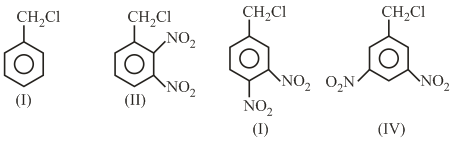
The decreasing order of reactivity of the following compounds towards nucleophilic substitution ($$S_N2$$) is:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 39
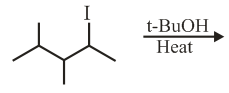
The major product in the following reaction is:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 40
Consider the following reaction:
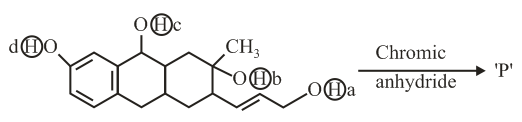
The product 'P' gives positive ceric ammonium nitrate test. This is because of the presence of which of these -OH group(s)?
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 41
The increasing order of the reactivity of the following compounds in nucleophile addition reaction is:
Propanal, Benzaldehyde, Propanone, Butanone
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 42
The compound A in the following reactions is:
$$A \xrightarrow[(ii)Conc. H_{2}SO_{4}/ \Delta]{(i)CH_{3}MgBr/H_{2}O} B \xrightarrow[(ii) Zn/H_{2}O]{(i) O_{3}} C + D$$
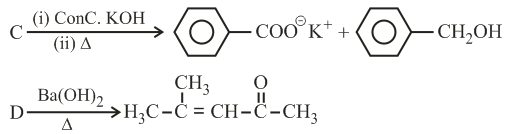
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 43
Three isomers A, B and C (mol. formula $$C_8H_{11}N$$) give the following results:
A and C $$\xrightarrow{Diazotization}$$ P + Q $$\xrightarrow[(ii) acidation(KMnO_4+H^+)]{(i) Hydrolysis}$$ R (product of A) + S (product of C)
R has lower boiling point than S
B $$\xrightarrow{C_6H_5SO_2Cl}$$ alkali-insoluble product
A, B and C, respectively are:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 44
An ionic micelle is formed on the addition of:
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 45
Match the following drugs with their therapeutic actions:
(i) Ranitidine (a) Antidepressant
(ii) Nardil (Phenelzine) (b) Antibiotic
(iii) Chloramphenicol (c) Antihistamine
(iv) Dimetane (Brompheniramine) (d) Antacid
(e) Analgesic
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 46
$$0.023 \times 10^{22}$$ molecules are present in 10g of a substance 'x'. The molarity of a solution containing 5g of substance 'x' in 2 L solution is _________ $$\times 10^{-3}$$
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 47
If 250 cm$$^3$$ of an aqueous solution containing 0.73g of a protein A is isotonic with one litre of another aqueous solution containing 1.65g of a protein B, at 298K, the ratio of the molecular masses of A and B is _________ $$\times 10^{-2}$$ (to the nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 48
An acidic solution of dichromate is electrolyzed for 8 minutes using 2 A current. As per the following equation $$Cr_2O_7^{2-} + 14H^+ + 6e^- \to 2Cr^{3+} + 7H_2O$$
The amount of $$Cr^{3+}$$ obtained was 0.104g. The efficiency of the process (in %) is (Take: F = 960000C, At. mass of chromium = 52)
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 49
The volume (in mL) of 0.1 N NaOH required to neutralise 10 mL of 0.1 N phosphinic acid is __________.
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 2 - Chemistry - Question 50
The number of
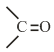
groups present in a tripeptide Asp - Glu - Lys is ____
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




