NTA JEE Main 3rd September 2020 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 26
The atomic number of the element unnilennium is:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 27
If the boiling point of $$H_2O$$ is 373 K, and the boiling point of $$H_2S$$ will be:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 28
Of the species, NO, NO$$^+$$, NO$$^{2+}$$ and NO$$^-$$, the one with minimum bond strength is:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 29
An acidic buffer is obtained on mixing:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 30
Which one of the following compounds possesses the most acidic hydrogen?
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 31
Glycerol is separated in soap industries by:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 32
The Kjeldahl method of Nitrogen estimation fails for which of the following reaction products?
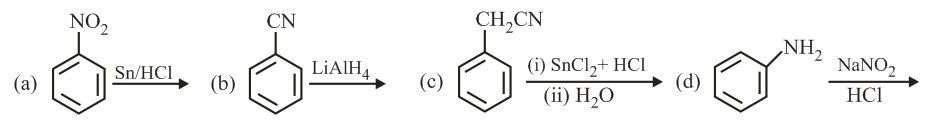
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 33
Which of the following compounds produces an optically inactive compound on hydrogenation?
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 34
Thermal power plants can lead to:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 35
Henry's constant (in kbar) for four gases $$\alpha$$, $$\beta$$, $$\gamma$$ and $$\delta$$ in water at 298 K is given below:
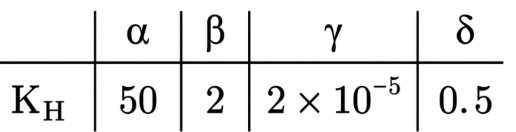
(density of water = 10$$^3$$ kg m$$^{-3}$$ at 298 K) This table implies that:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 36
Let $$C_{NaCl}$$ and $$C_{BaSO_4}$$, be the conductances (in S) measured for saturated aqueous solutions of NaCl and $$BaSO_4$$ respectively, at a temperature T. Which of the following is false?
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 37
It is true that:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 38
Tyndall effect is observed when:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 39
Aqua regia is used for dissolving noble metals (Au, Pt, etc.). The gas evolved in this process is
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 40
In a molecule of pyrophosphoric acid, the number of P - O - H, P = O and P - O - P bonds/moiety(ies) respectively are:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 41
The electronic spectrum of $$\left[Ti(H_2O)_6\right]^{3+}$$ shows a single broad peak with a maximum at 20,300 cm$$^{-1}$$. The crystal field stabilization energy (CFSE) of the complex ion, in kJ mol$$^{-1}$$, is: (1 kJ mol$$^{-1}$$ = 83.7 cm$$^{-1}$$)
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 42
The complex that can show optical activity is:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 43
The mechanism of Sn1 reaction is given as:
$$R - X \rightarrow R^\oplus X^\ominus \rightarrow R^\oplus \| X^\ominus \xrightarrow{Y^\ominus} R - Y + X^\ominus$$
Ion pair Solvent separated ion pair
A student writes general characteristics based on the given mechanism as:
(a) The reaction is favoured by weak nucleophiles.
(b) $$R^\oplus$$ would be easily formed if the substituents are bulky
(c) The reaction is accompanied by racemization
(d) The reaction is favoured by non-polar solvents.
Which observations are correct?
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 44
An organic compound [A], molecular formula $$C_{10}H_{20}O_2$$ was hydrolyzed with dilute sulphuric acid to give a carboxylic acid [B] and an alcohol [C]. Oxidation of [C] with $$CrO_3 - H_2SO_4$$ produced [B]. Which of the following structures are not possible for [A]?
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 45
The antifertility drug "Novestrol" can react with:
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 46
The mole fraction of glucose ($$C_6H_{12}O_6$$) in an aqueous binary solution is 0.1. The mass percentage of water in it, to the nearest integer, is .........
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 47
The volume strength of 8.9 M $$H_2O_2$$ solution calculated at 273 K and 1 atm is ......... ($$R = 0.0821$$ L atm K$$^{-1}$$ mol$$^{-1}$$) (rounded off to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 48
The total number of monohalogenated organic products in the following (including stereoisomers) reaction is
A (Simplest optically active alkene) $$\xrightarrow[(ii) X_2/\Delta]{(i) H_2/Ni/\Delta}$$
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 49
An element with molar mass $$2.7 \times 10^{-2}$$ kg mol$$^{-1}$$ forms a cubic unit cell with edge length 405 pm. If its density is $$2.7 \times 10^3$$ kg m$$^{-3}$$, the radius of the element is approximately ......... $$\times 10^{-12}$$ m (to the nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 3rd September 2020 Shift 1 - Chemistry - Question 50
The photoelectric current from Na (work function, $$w_0 = 2.3$$ eV) is stopped by the output voltage of the cell
Pt(s)|H$$_2$$(g, 1 bar)|HCl(aq, pH = 1)|AgCl(s)|Ag(s)
The pH of aq. HCl required to stop the photoelectric current from K($$w_0 = 2.25$$ eV), all other conditions remaining the same, is ......... $$\times 10^{-2}$$ (to the nearest integer).
Given $$2.303\frac{RT}{F} = 0.06$$ V; $$E^0_{AgCl/Ag/Cl} = 0.22$$ V
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




