NTA JEE Main 29th June 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 31
Using the rules for significant figures, the correct answer for the expression $$\frac{0.02858 \times 0.112}{0.5702}$$ will be:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 32
Which of the following is the correct plot for the probability density $$\psi^2(r)$$ as a function of distance 'r' of the electron form the nucleus for 2s orbital?
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 33
Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: The first ionization enthalpy for oxygen is lower than that of nitrogen.
Reason R: The four electrons in 2p orbitals of oxygen experience more electron-electron repulsion.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 34
Consider the species CH$$_4$$, NH$$_4^+$$ and BH$$_4^-$$. Choose the correct option with respect to the three species:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 35
Number of lone pair (s) of electrons on central atom and the shape of BrF$$_3$$ molecule respectively, are:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 36
4.0 moles of argon and 5.0 moles of PCl$$_5$$ are introduced into an evacuated flask of 100 litre capacity at 610 K. The system is allowed to equilibrate. At equilibrium, the total pressure of mixture was found to be 6.0 atm. The K$$_p$$ for the reaction is [Given: R = 0.082 L atm K$$^{-1}$$ mol$$^{-1}$$]
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 37
Amongst baking soda, caustic soda and washing soda carbonate anion is present in:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 38
Aqueous solution of which of the following boron compounds will be strongly basic in nature?
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 39
Which of the following carbocations is most stable?
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 40
Sulphur dioxide is one of the components of polluted air. SO$$_2$$ is also a major contributor to acid rain. The correct and complete reaction to represent acid rain caused by SO$$_2$$ is:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 41
42.12% (w/v) solution of NaCl causes precipitation of a certain sol in 10 hours. The coagulating value of NaCl for the sol is
[Given: Molar mass: Na = 23.0 g mol$$^{-1}$$; Cl = 35.5 g mol$$^{-1}$$]
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 42
Match List I with List II.
List I With Ore List II Composition
(A) Siderite (I) FeCO$$_3$$
(B) Malachite (II) CuCO$$_3$$ · Cu(OH)$$_2$$
(C) Sphalerite (III) ZnS
(D) Calamine (IV) ZnCO$$_3$$
Choose the correct answer from the options given below:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 43
Given below are two statements.
Statement I: In CuSO$$_4$$ · 5H$$_2$$O, Cu - O bonds are present.
Statement II: In CuSO$$_4$$ · 5H$$_2$$O, ligands coordinating with Cu (II) ion are O-and S-based ligands.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 44
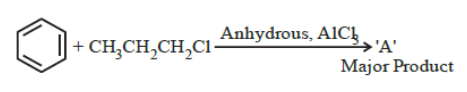
The stable carbocation formed in the above reaction is:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 45
Two isomers (A) and (B) with Molar mass 184 g/mol and elemental composition C, 52.2%; H, 49% and Br 42.9% gave benzoic acid and p-bromobenzoic acid, respectively on oxidation with KMnO$$_4$$. Isomer 'A' is optically active and gives a pale yellow precipitate when warmed with alcoholic AgNO$$_3$$. Isomer 'A' and 'B' are, respectively:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 46
In Friedel-Crafts alkylation of aniline, one gets:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 47
Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Dacron is an example of polyester polymer.
Reason R: Dacron is made up of ethylene glycol and terephthalic acid monomers.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 48
The mixture of chloroxylenol and terpineol is an example of:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 49
A white precipitate was formed when BaCl$$_2$$ was added to water extract of an inorganic salt. Further, a gas 'X' with characteristic odour was released when the formed white precipitate was dissolved in dilute HCI. The anion present in the inorganic salt is:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 50
The structure of protein that is unaffected by heating is:
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 51
A box contains 0.90 g of liquid water in equilibrium with water vapour at 27°C. The equilibrium vapour pressure of water at 27°C 32.0 Torr. When the volume of the box is increased, some of the liquid water evaporates to maintain the equilibrium pressure. If all the liquid water evaporates, then the volume of the box must be ______ litre. [nearest integer]
(Given: R = 0.082 L atm K$$^{-1}$$ mol$$^{-1}$$)
(Ignore the volume of the liquid water and assume water vapours behave as an ideal gas.)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 52
2.2 g of nitrous oxide (N$$_2$$O) gas is cooled at a constant pressure of 1 atm from 310 K to 270 K causing the compression of the gas from 217.1 mL to 167.75 mL. The change in internal energy of the process, $$\Delta$$U is '-x'J. The value of 'x' is ______.
[nearest integer]
(Given: atomic mass of N = 14 g mol$$^{-1}$$ and of O = 16 g mol$$^{-1}$$. Molar heat capacity of N$$_2$$O is 100 JK$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 53
For the reaction given below:
CoCl$$_3$$ · xNH$$_3$$ + AgNO$$_3$$(aq) $$\rightarrow$$
If two equivalents of AgCl precipitate out, then the value of x will be ______.
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 54
The number of chiral alcohol(s) with molecular formula C$$_4$$H$$_{10}$$O is ______ (Assume stereoisomers as different chiral alcohols)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 55
Elevation in boiling point for 1.5 molal solution of glucose in water is 4 K. The depression in freezing point for 4.5 molal solution of glucose in water is 4 K. The ratio of molal elevation constant to molal depression constant (K$$_b$$/K$$_f$$) is
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 56
The cell potential for the given cell at 298 K Pt|H$$_2$$(g, 1 bar)|H$$^+$$(aq)||Cu$$^{2+}$$(aq)|Cu(s) is 0.31 V. The pH of the acidic solution is found to be 3, whereas the concentration of Cu$$^{2+}$$ is $$10^x$$ M. The value of x is ______.
(Given: $$E^\ominus_{Cu^{2+}/Cu} = 0.34$$ V and $$\frac{2.303RT}{F} = 0.06$$ V)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 57
The equation $$k = (6.5 \times 10^{12} s^{-1})e^{-26000 K/T}$$ is followed for the decomposition of compound A. The activation energy for the reaction is ______ KJ mol$$^{-1}$$. [nearest integer]
(Given: R = 8.314 J K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 58
Spin only magnetic moment of $$[MnBr_6]^{4-}$$ is ______ B.M. (round off to the closest integer)
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 59
In the given reaction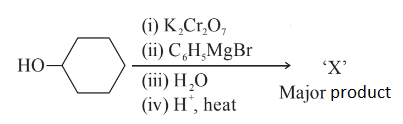
the number of sp$$^2$$ hybridised carbon (s) in compound 'X' is ______.
789
456
123
0.-
Clear All
NTA JEE Main 29th June 2022 Shift 2 - Chemistry - Question 60
In the given reaction,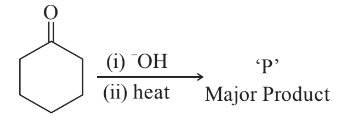
The number of $$\pi$$ electrons present in the product 'P' is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




