NTA JEE Main 29th July 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 29th July 2022 Shift 1 - Question 41
In following pairs, the one in which both transition metal ions are colourless is
NTA JEE Main 29th July 2022 Shift 1 - Question 42
In neutral or faintly alkaline medium, $$KMnO_4$$ being a powerful oxidant can oxidise, thiosulphate almost quantitatively, to sulphate. In this reaction overall change in oxidation state of manganese will be
NTA JEE Main 29th July 2022 Shift 1 - Question 43
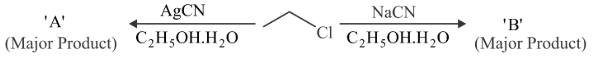
Considering the above reactions, the compound 'A' and compound 'B' respectively are
NTA JEE Main 29th July 2022 Shift 1 - Question 44
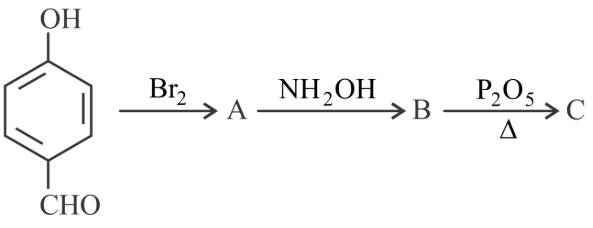
Consider the above reaction sequence, the Product 'C' is
NTA JEE Main 29th July 2022 Shift 1 - Question 45
A compound 'X' is acidic and it is soluble in NaOH solution, but insoluble in $$NaHCO_3$$ solution. Compound 'X' also gives violet colour with neutral $$FeCl_3$$ solution. The compound 'X' is
NTA JEE Main 29th July 2022 Shift 1 - Question 46
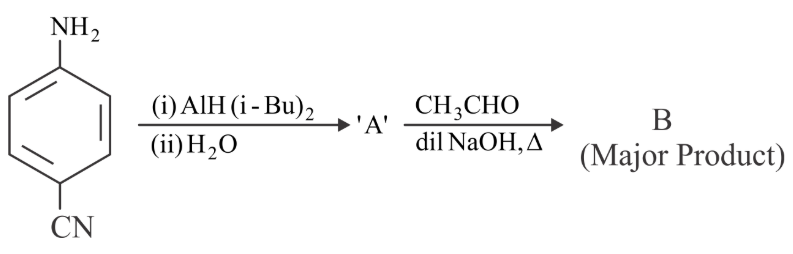
Consider the following reaction sequence. The product 'B' is
NTA JEE Main 29th July 2022 Shift 1 - Question 47
Which among the following is the strongest Bronsted base?
NTA JEE Main 29th July 2022 Shift 1 - Question 48
Consider the above reaction, the compound 'A' is
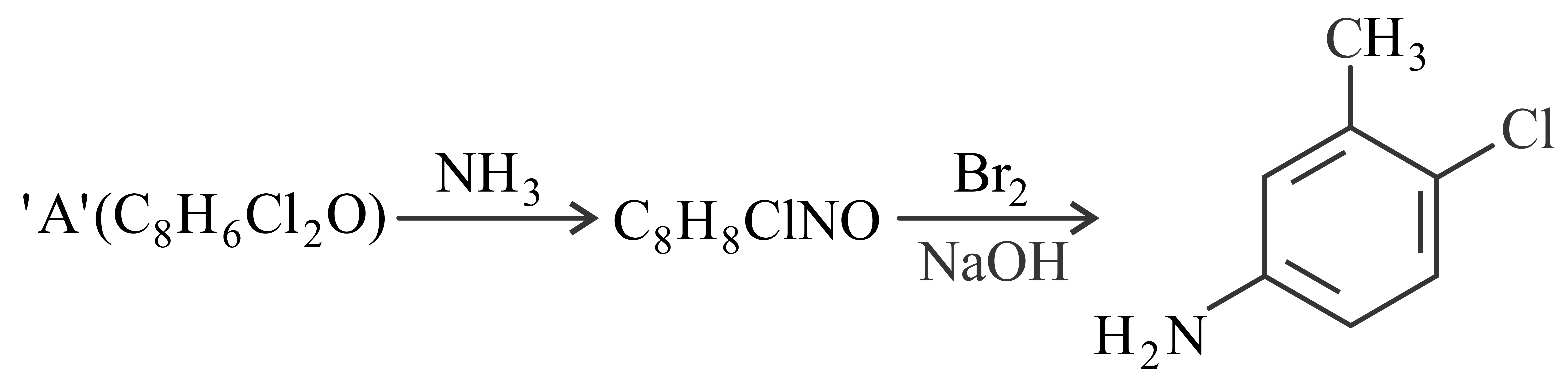
NTA JEE Main 29th July 2022 Shift 1 - Question 49
Which among the following represent reagent 'A'?
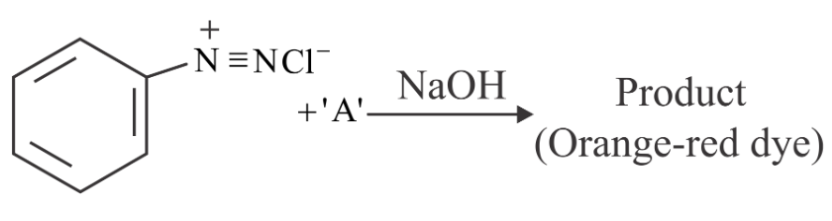
NTA JEE Main 29th July 2022 Shift 1 - Question 50
Which of the following compounds is an example of hypnotic drug?


