NTA JEE Main 29th January 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 31
Given below are two statements:
Statement I: The decrease in first ionization enthalpy from B to Al is much larger than that from Al to Ga.
Statement II: The d orbitals in Ga are completely filled.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 32
According to MO theory the bond orders for O$$_2^{2-}$$, CO and NO$$^+$$ respectively, are
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 33
Which of the following relations are correct?
(A) $$\Delta U = q + p\Delta V$$
(B) $$\Delta G = \Delta H - T\Delta S$$
(C) $$\Delta S = \frac{q_{rev}}{T}$$
(D) $$\Delta H = \Delta U - \Delta nRT$$
Choose the most appropriate answer from the options given below:
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 34
An indicator 'X' is used for studying the effect of variation in concentration of iodide on the rate of reaction of iodide ion with H$$_2$$O$$_2$$ at room temp. The indicator 'X' forms blue colored complex with compound 'A' present in the solution. The indicator 'X' and compound 'A' respectively are
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 35
Given below are two statements:
Statement I: Nickel is being used as the catalyst for producing syn gas and edible fats.
Statement II: Silicon forms both electron rich and electron deficient hydrides.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 36
When a hydrocarbon A undergoes combustion in the presence of air, it requires $$9.5$$ equivalents of oxygen and produces $$3$$ equivalents of water. What is the molecular formula of A?
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 37
The concentration of dissolved Oxygen in water for growth of fish should be more than X ppm and Biochemical Oxygen Demand in clean water should be less than Y ppm. X and Y in ppm are, respectively.
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 38
Match List I with List II.
| List I | List II |
|---|---|
| A. van't Hoff factor, i | I. Cryoscopic constant |
| B. k$$_f$$ | II. Isotonic solutions |
| C. Solutions with same osmotic pressure | III. $$\frac{\text{Normal molar mass}}{\text{Abnormal molar mass}}$$ |
| D. Azeotropes | IV. Solutions with same composition of vapour above it |
Choose the correct answer from the options given below:
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 39
Match List-I and List-II
| List-I | List-II |
|---|---|
| A. Osmosis | I. Solvent molecules pass through semi permeable membrane towards solvent side. |
| B. Reverse osmosis | II. Movement of charged colloidal particles under the influence of applied electric potential towards oppositely charged electrodes |
| C. Electro osmosis | III. Solvent molecules pass through semi permeable membrane towards solution side |
| D. Electrophoresis | IV. Dispersion medium moves in an electric field. |
Choose the correct answer from the options given below:
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 40
The major component of which of the following ore is sulphide based mineral?
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 41
A solution of CrO$$_5$$ in amyl alcohol has a ____colour
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 42
The set of correct statements is:
(i) Manganese exhibits $$+7$$ oxidation state in its oxide.
(ii) Ruthenium and Osmium exhibit $$+8$$ oxidation in their oxides.
(iii) Sc shows $$+4$$ oxidation state which is oxidizing in nature.
(iv) Cr shows oxidising nature in $$+6$$ oxidation state.
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 43
Correct order of spin only magnetic moment of the following complex ions is:
(Given At. No. Fe : 26, Co : 27)
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 44
The one giving maximum number of isomeric alkenes on dehydrohalogenation reaction is (excluding rearrangement)
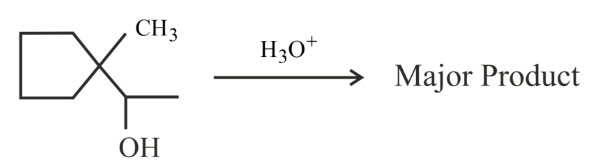
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 45
Find out the major product for the following reaction
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 46
Find out the major products from the following reaction sequence.
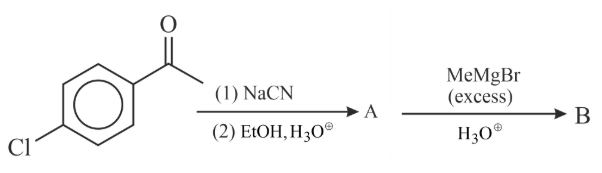
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 47
Reaction of propanamide with Br$$_2$$ / KOH (aq) produces:
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 48
Match List-I and List-II.
| List-I | List-II |
|---|---|
| A. Elastomeric polymer | I. Urea formaldehyde resin |
| B. Fibre polymer | II. Polystyrene |
| C. Thermosetting polymer | III. Polyester |
| D. Thermoplastic polymer | IV. Neoprene |
Choose the correct answer from the options given below:
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 49
A doctor prescribed the drug Equanil to a patient. The patient was likely to have symptoms of which disease?
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 50
Following tetrapeptide can be represented as
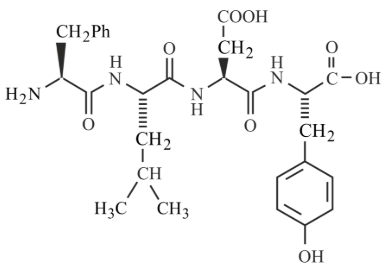
(F, L, D, Y, I, Q, P are one letter codes for amino acids)
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 51
Assume that the radius of the first Bohr orbit of hydrogen atom is $$0.6$$ $$\text{\AA}$$. The radius of the third Bohr orbit of He$$^+$$ is ______ picometer. (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 52
At 298 K
$$\text{N}_2(g) + 3\text{H}_2(g) \rightleftharpoons 2\text{NH}_3(g), K_1 = 4 \times 10^5$$
$$\text{N}_2(g) + \text{O}_2(g) \rightleftharpoons 2\text{NO}(g), K_2 = 1.6 \times 10^{12}$$
$$\text{H}_2(g) + \frac{1}{2}\text{O}_2(g) \rightleftharpoons \text{H}_2\text{O}(g), K_3 = 1.0 \times 10^{-13}$$
Based on above equilibria, the equilibrium constant of the reaction,
$$2\text{NH}_3(g) + \frac{5}{2}\text{O}_2(g) \rightleftharpoons 2\text{NO}(g) + 3\text{H}_2\text{O}(g)$$
is ______ $$\times 10^{-33}$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 53
The volume of HCl, containing $$73$$ g L$$^{-1}$$, required to completely neutralise NaOH obtained by reacting $$0.69$$ g of metallic sodium with water, is ______ mL. (Nearest Integer)
(Given: molar Masses of Na, Cl, O, H are 23, 35.5, 16 and 1 g mol$$^{-1}$$ respectively)
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 54
On heating, LiNO$$_3$$ gives how many compounds among the following?
Li$$_2$$O, N$$_2$$, O$$_2$$, LiNO$$_2$$, NO$$_2$$
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 55
When $$0.01$$ mol of an organic compound containing $$60\%$$ carbon was burnt completely, $$4.4$$ g of CO$$_2$$ was produced. The molar mass of compound is ______ g mol$$^{-1}$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 56
A metal M forms hexagonal close-packed structure. The total number of voids in $$0.02$$ mol of it is ______ $$\times 10^{21}$$ (Nearest integer)
(Given N$$_A = 6.02 \times 10^{23}$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 57
The equilibrium constant for the reaction Zn(s) + Sn$$^{2+}$$(aq) $$\rightleftharpoons$$ Zn$$^{2+}$$(aq) + Sn(s) is $$1 \times 10^{20}$$ at 298 K. The magnitude of standard electrode potential of Sn/Sn$$^{2+}$$ if E$$^0_{\text{Zn}^{2+}/\text{Zn}} = -0.76$$ V is ______ $$\times 10^{-2}$$ V. (Nearest integer)
Given: $$\frac{2.303 RT}{F} = 0.059$$ V
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 58
For conversion of compound A $$\to$$ B, the rate constant of the reaction was found to be $$4.6 \times 10^{-5}$$ L mol$$^{-1}$$ s$$^{-1}$$. The order of the reaction is ______.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 59
Total number of acidic oxides among N$$_2$$O$$_3$$, NO$$_2$$, N$$_2$$O, Cl$$_2$$O$$_7$$, SO$$_2$$, CO, CaO, Na$$_2$$O and NO is ______.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 2 - Chemistry - Question 60
The denticity of the ligand present in the Fehling's reagent is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




