NTA JEE Main 29th January 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 29th January 2023 Shift 1 - Question 51
Number of cyclic tripeptides formed with 2 amino acids A and B is:
NTA JEE Main 29th January 2023 Shift 1 - Question 52
The number of molecules or ions from the following, which do not have odd number of electrons are ______.
(A) NO$$_2$$
(B) ICl$$_4^-$$
(C) BrF$$_3$$
(D) ClO$$_2$$
(E) NO$$_2^+$$
(F) NO
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 53
Consider the following reaction approaching equilibrium at $$27°$$C and 1 atm pressure
$$\text{A} + \text{B} \underset{K_r=10^2}{\overset{K_f=10^3}{\rightleftharpoons}} \text{C} + \text{D}$$
The standard Gibb's energy change $$(\Delta_r G°)$$ at $$27°$$C is $$(-)$$ ______ kJ mol$$^{-1}$$. (Nearest integer).
(Given: R $$= 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$ and $$\ln 10 = 2.3$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 54
Water decomposes at 2300 K
$$\text{H}_2\text{O}(g) \to \text{H}_2(g) + \frac{1}{2}\text{O}_2(g)$$
The percent of water decomposing at 2300 K and 1 bar is ______ (Nearest integer). Equilibrium constant for the reaction is $$2 \times 10^{-3}$$ at 2300 K
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 55
Millimoles of calcium hydroxide required to produce 100 mL of the aqueous solution of pH 12 is $$x \times 10^{-1}$$. The value of $$x$$ is ______ (Nearest integer). Assume complete dissociation.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 56
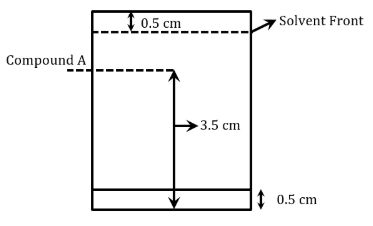
Following chromatogram was developed by adsorption of compound 'A' on a 6 cm TLC glass plate. Retardation factor of the compound 'A' is ______ $$\times 10^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 57
17 mg of a hydrocarbon (M.F. C$$_{10}$$H$$_{16}$$) takes up 8.40 mL of the H$$_2$$ gas measured at 0°C and 760 mm of Hg. Ozonolysis of the same hydrocarbon yields
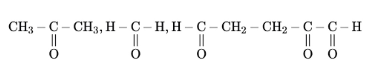
The number of double bond/s present in the hydrocarbon is
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 58
Solid Lead nitrate is dissolved in 1 litre of water. The solution was found to boil at $$100.15°$$C. When $$0.2$$ mol of NaCl is added to the resulting solution, it was observed that the solution froze at $$-0.8°$$C. The solutibility product of PbCl$$_2$$ formed is ______ $$\times 10^{-6}$$ at 298 K. (Nearest integer)
Given: K$$_b = 0.5$$ K kg mol$$^{-1}$$ and K$$_f = 1.8$$ kg mol$$^{-1}$$. Assume molality to be equal to molarity in all cases.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 59
For certain chemical reaction X $$\to$$ Y, the rate of formation of product is plotted against the time as shown in the figure. The number of Correct statement/s from the following is ______
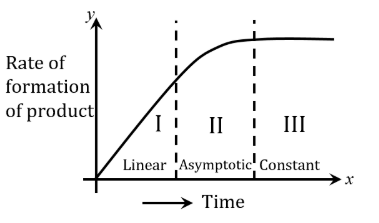
(A) Over all order of this reaction is one
(B) Order of this reaction can't be determined
(C) In region-I and III, the reaction is of first and zero order respectively
(D) In region-II, the reaction is of first order
(E) In region-II, the order of reaction is in the range of 0.1 to 0.9.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Question 60
The sum of bridging carbonyls in W(CO)$$_6$$ and Mn$$_2$$(CO)$$_{10}$$ is ______.
789
456
123
0.-
Clear All


