NTA JEE Main 29th January 2023 Shift 1
For the following questions answer them individually
NTA JEE Main 29th January 2023 Shift 1 - Question 31
The shortest wavelength of hydrogen atom in Lyman series is $$\lambda$$. The longest wavelength in Balmer series of He$$^+$$ is
NTA JEE Main 29th January 2023 Shift 1 - Question 32
The bond dissociation energy is highest for
NTA JEE Main 29th January 2023 Shift 1 - Question 33
For 1 mol of gas, the plot of pV vs p is shown below. p is the pressure and V is the volume of the gas.
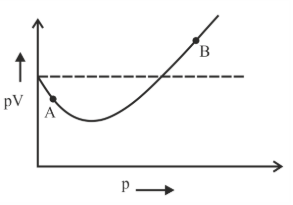
What is the value of compressibility factor at point A?
NTA JEE Main 29th January 2023 Shift 1 - Question 34
Which of the given compounds can enhance the efficiency of hydrogen storage tank?
NTA JEE Main 29th January 2023 Shift 1 - Question 35
The magnetic behaviour of Li$$_2$$O, Na$$_2$$O$$_2$$ and KO$$_2$$, respectively, are
NTA JEE Main 29th January 2023 Shift 1 - Question 36
The correct order of hydration enthalpies is
(A) K$$^+$$
(B) Rb$$^+$$
(C) Mg$$^{2+}$$
(D) Cs$$^+$$
(E) Ca$$^{2+}$$
Choose the correct answer from the options below:
NTA JEE Main 29th January 2023 Shift 1 - Question 37
During the borax bead test with CuSO$$_4$$, a blue green colour of the bead was observed in oxidising flame due to the formation of
NTA JEE Main 29th January 2023 Shift 1 - Question 38
Compound that will give positive Lassaigne's test for both nitrogen and halogen is
NTA JEE Main 29th January 2023 Shift 1 - Question 39
Correct statement about smog is
NTA JEE Main 29th January 2023 Shift 1 - Question 40
The standard electrode potential (M$$^{3+}$$/M$$^{2+}$$) for V, Cr, Mn & Co are $$-0.26$$ V, $$-0.41$$ V, $$+1.57$$ V and $$+1.97$$ V, respectively. The metal ions which can liberate H$$_2$$ from a dilute acid are


