NTA JEE Main 29th January 2023 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 31
The shortest wavelength of hydrogen atom in Lyman series is $$\lambda$$. The longest wavelength in Balmer series of He$$^+$$ is
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 32
The bond dissociation energy is highest for
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 33
For 1 mol of gas, the plot of pV vs p is shown below. p is the pressure and V is the volume of the gas.
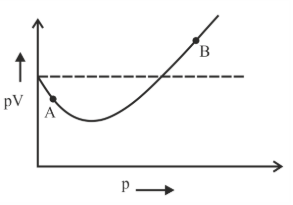
What is the value of compressibility factor at point A?
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 34
Which of the given compounds can enhance the efficiency of hydrogen storage tank?
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 35
The magnetic behaviour of Li$$_2$$O, Na$$_2$$O$$_2$$ and KO$$_2$$, respectively, are
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 36
The correct order of hydration enthalpies is
(A) K$$^+$$
(B) Rb$$^+$$
(C) Mg$$^{2+}$$
(D) Cs$$^+$$
(E) Ca$$^{2+}$$
Choose the correct answer from the options below:
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 37
During the borax bead test with CuSO$$_4$$, a blue green colour of the bead was observed in oxidising flame due to the formation of
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 38
Compound that will give positive Lassaigne's test for both nitrogen and halogen is
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 39
Correct statement about smog is
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 40
The standard electrode potential (M$$^{3+}$$/M$$^{2+}$$) for V, Cr, Mn & Co are $$-0.26$$ V, $$-0.41$$ V, $$+1.57$$ V and $$+1.97$$ V, respectively. The metal ions which can liberate H$$_2$$ from a dilute acid are
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 41
Following figure shows dependence of molar conductance of two electrolytes on concentration. $$\Lambda^0_m$$ is the limiting molar conductivity.
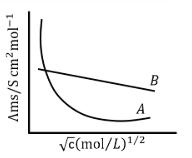
The number of Incorrect statement(s) from the following is ______
(A) $$\Lambda^0_m$$ for electrolyte A is obtained by extrapolation
(B) For electrolyte B, $$\Lambda_m$$ Vs $$\sqrt{c}$$ graph is a straight line with intercept equal to $$\Lambda^0_m$$
(C) At infinite dilution, the value of degree of dissociation approach zero for electrolyte B.
(D) $$\Lambda_m$$ for any electrolyte A or B can be calculated using $$\lambda°$$ for individual ions.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 42
Which of the following salt solutions would coagulate the colloid solution formed when FeCl$$_3$$ is added to NaOH solution, at the fastest rate?
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 43
The reaction representing the Mond process for metal refining is ______
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 44
"A" obtained by Ostwald's method involving air oxidation of NH$$_3$$, upon further air oxidation produces "B". "B" on hydration forms an oxoacid of Nitrogen along with evolution of "A". The oxoacid also produces "A" and gives positive brown ring test
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 45
Chiral complex from the following is:
Here en = ethylene diamine
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 46
Identify the correct order for the given property for following compounds
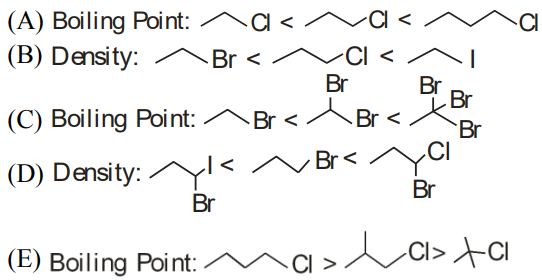
Choose the correct answer from the option below:
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 47
The increasing order of pK$$_a$$ for the following phenols is
(A) 2,4-Dinitrophenol
(B) 4-Nitrophenol
(C) 2,4,5-Trimethylphenol
(D) Phenol
(E) 3-Chlorophenol
Choose the correct answer from the option given below:
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 48
Match List I with List II.
| List I (Reaction) | List II (Reagents) |
|---|---|
| (A) Hoffmann Degradation | (I) Conc. KOH, $$\Delta$$ |
| (B) Clemmensen reduction | (II) CHCl$$_3$$, NaOH/H$$_3$$O$$^+$$ |
| (C) Cannizzaro reaction | (III) Br$$_2$$, NaOH |
| (D) Reimer-Tiemann reaction | (IV) Zn $$-$$ Hg/HCl |
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 49
The major product 'P' for the following sequence of reactions is:
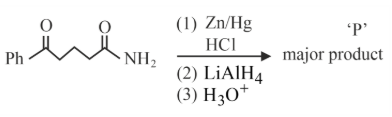
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 50
Match List I with List II
| List I (Antimicrobials) | List II (Names) |
|---|---|
| (A) Narrow Spectrum Antibiotic | (I) Furacin |
| (B) Antiseptic | (II) Sulphur dioxide |
| (C) Disinfectants | (III) Penicillin-G |
| (D) Broad spectrum antibiotic | (IV) Chloramphenicol |
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 51
Number of cyclic tripeptides formed with 2 amino acids A and B is:
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 52
The number of molecules or ions from the following, which do not have odd number of electrons are ______.
(A) NO$$_2$$
(B) ICl$$_4^-$$
(C) BrF$$_3$$
(D) ClO$$_2$$
(E) NO$$_2^+$$
(F) NO
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 53
Consider the following reaction approaching equilibrium at $$27°$$C and 1 atm pressure
$$\text{A} + \text{B} \underset{K_r=10^2}{\overset{K_f=10^3}{\rightleftharpoons}} \text{C} + \text{D}$$
The standard Gibb's energy change $$(\Delta_r G°)$$ at $$27°$$C is $$(-)$$ ______ kJ mol$$^{-1}$$. (Nearest integer).
(Given: R $$= 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$ and $$\ln 10 = 2.3$$)
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 54
Water decomposes at 2300 K
$$\text{H}_2\text{O}(g) \to \text{H}_2(g) + \frac{1}{2}\text{O}_2(g)$$
The percent of water decomposing at 2300 K and 1 bar is ______ (Nearest integer). Equilibrium constant for the reaction is $$2 \times 10^{-3}$$ at 2300 K
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 55
Millimoles of calcium hydroxide required to produce 100 mL of the aqueous solution of pH 12 is $$x \times 10^{-1}$$. The value of $$x$$ is ______ (Nearest integer). Assume complete dissociation.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 56
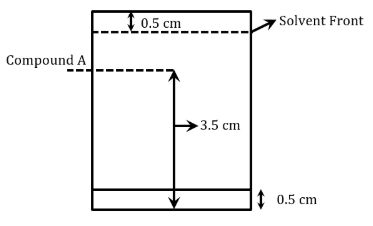
Following chromatogram was developed by adsorption of compound 'A' on a 6 cm TLC glass plate. Retardation factor of the compound 'A' is ______ $$\times 10^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 57
17 mg of a hydrocarbon (M.F. C$$_{10}$$H$$_{16}$$) takes up 8.40 mL of the H$$_2$$ gas measured at 0°C and 760 mm of Hg. Ozonolysis of the same hydrocarbon yields
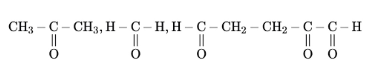
The number of double bond/s present in the hydrocarbon is
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 58
Solid Lead nitrate is dissolved in 1 litre of water. The solution was found to boil at $$100.15°$$C. When $$0.2$$ mol of NaCl is added to the resulting solution, it was observed that the solution froze at $$-0.8°$$C. The solutibility product of PbCl$$_2$$ formed is ______ $$\times 10^{-6}$$ at 298 K. (Nearest integer)
Given: K$$_b = 0.5$$ K kg mol$$^{-1}$$ and K$$_f = 1.8$$ kg mol$$^{-1}$$. Assume molality to be equal to molarity in all cases.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 59
For certain chemical reaction X $$\to$$ Y, the rate of formation of product is plotted against the time as shown in the figure. The number of Correct statement/s from the following is ______
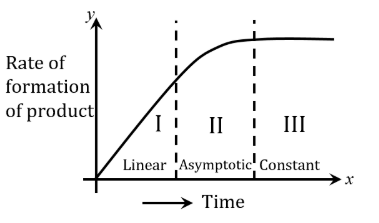
(A) Over all order of this reaction is one
(B) Order of this reaction can't be determined
(C) In region-I and III, the reaction is of first and zero order respectively
(D) In region-II, the reaction is of first order
(E) In region-II, the order of reaction is in the range of 0.1 to 0.9.
789
456
123
0.-
Clear All
NTA JEE Main 29th January 2023 Shift 1 - Chemistry - Question 60
The sum of bridging carbonyls in W(CO)$$_6$$ and Mn$$_2$$(CO)$$_{10}$$ is ______.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




