NTA JEE Main 28th June 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 31
Compound A contains 8.7% Hydrogen, 74% Carbon and 17.3% Nitrogen. The molecular formula of the compound is, Given: Atomic masses of C, H and N are 12, 1 and 14 amu respectively. The molar mass of the compound A is 162 g mol$$^{-1}$$.
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 32
Consider the following statements:
(A) The principal quantum number 'n' is a positive integer with values of 'n' = 1, 2, 3, ...
(B) The azimuthal quantum number 'l' for a given 'n' (principal quantum number) can have values as 'l' = 0, 1, 2, ... n has $$(2n + 1)$$ values.
(C) Magnetic orbital quantum number 'm' for a particular 'l' (azimuthal quantum number) has $$(2l + 1)$$ values.
(D) $$\pm \frac{1}{2}$$ are the two possible orientations of electron spin.
(E) For $$l = 5$$, there will be a total of 9 orbital.
Which of the above statements are correct?
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 33
Match List-I with List-II
List-I List-II
(A) Cl$$_2$$O$$_7$$ (I) Amphoteric
(B) Na$$_2$$O (II) Basic
(C) Al$$_2$$O$$_3$$ (III) Neutral
(D) N$$_2$$O (IV) Acidic
Choose the correct answer from the options given below
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 34
In the structure of SF$$_4$$, the lone pair of electrons on S is in.
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 35
A student needs to prepare a buffer solution of propanoic acid and its sodium salt with pH 4. The ratio of $$\frac{[CH_3CH_2COO^-]}{[CH_3CH_2COOH]}$$ required to make buffer is Given: $$K_a(CH_3CH_2COOH) = 1.3 \times 10^{-5}$$
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 36
Hydrogen has three isotopes: protium ($$^1$$H), deuterium ($$^2$$H or D) and tritium ($$^3$$H or T). They have nearly same chemical properties but different physical properties. They differ in
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 37
Among the following, basic oxide is
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 38
The correct IUPAC name of the following compound is
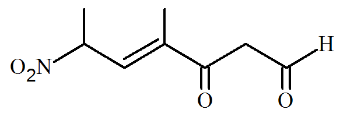
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 39
Correct statement about photo-chemical smog is
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 40
Match List-I with List-II
List-I List-II
(A) negatively charged sol (I) Fe$$_2$$O$$_3$$ · xH$$_2$$O
(B) macromolecular colloid (II) CdS sol
(C) positively charged sol (III) Starch
(D) Cheese (IV) a gel
Choose the correct answer from the options given below
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 41
In the metallurgical extraction of copper, following reaction is used:
$$\text{FeO} + \text{SiO}_2 \rightarrow \text{FeSiO}_3$$
FeO and FeSiO$$_3$$ respectively are.
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 42
Among the given oxides of nitrogen; N$$_2$$O, N$$_2$$O$$_3$$, N$$_2$$O$$_4$$ and N$$_2$$O$$_5$$, the number of compound/(s) having N - N bond is
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 43
Which of the following oxoacids of sulphur contains "S" in two different oxidation states?
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 44
Consider the following reaction,
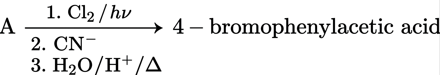
What is A in the above reaction?
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 45
The major product P of the given reaction is (where, Me is $$-$$CH$$_3$$)
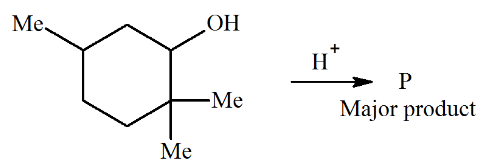
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 46
Isobutyraldehyde on reaction with formaldehyde and K$$_2$$CO$$_3$$ gives compound 'A'. Compound 'A' reacts with KCN and yields compound 'B', which on hydrolysis gives a stable compound 'C'. The compound 'C' is
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 47
With respect to the following reaction, consider the given statements:
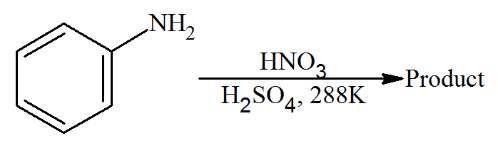
(A) o-Nitroaniline and p-nitroaniline are the predominant products.
(B) p-Nitroaniline and m-nitroaniline are the predominant products.
(C) HNO$$_3$$ acts as an acid.
(D) H$$_2$$SO$$_4$$ acts as an acid.
Choose the correct option.
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 48
Given below are two statements, one is Assertion and other is Reason.
Assertion: Natural rubber is a linear polymer of isoprene called cis-polyisoprene with elastic properties.
Reason: The cis-polyisoprene molecules consist of various chains held together by strong polar interactions with coiled structure.
In the light of the above statements, choose the correct one from the options given below
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 49
The structure of Tagamet (Cimetidine) is:
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 50
When sugar 'X' is boiled with dilute H$$_2$$SO$$_4$$ in alcoholic solution, two isomers 'A' and 'B' are formed. 'A' on oxidation with HNO$$_3$$ yields saccharic acid where as 'B' is laevorotatory. The compound 'X' is
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 51
The complete combustion of 0.492 g of an organic compound containing 'C', 'H' and 'O' gives 0.793 g of CO$$_2$$ and 0.442 g of H$$_2$$O. The percentage of oxygen composition in the organic compound is ______ (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 52
100 g of an ideal gas is kept in a cylinder of 416 L volume at 27°C under 1.5 bar pressure. The molar mass of the gas is ______ g mol$$^{-1}$$. (Nearest integer)
(Given: R = 0.083 L bar K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 53
For combustion of one mole of magnesium in an open container at 300 K and 1 bar pressure, $$\Delta_C H^\ominus = -601.70$$ kJ mol$$^{-1}$$, the magnitude of change in internal energy for the reaction is ______ kJ. (Nearest integer)
(Given: R = 8.3 J K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 54
0.01 M KMnO$$_4$$ solution was added to 20.0 mL of 0.05 M Mohr's salt solution through a burette. The initial reading of 50 mL burette is zero. The volume of KMnO$$_4$$ solution left in the burette after the end point is ______ mL. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 55
For the given reactions
$$Sn^{2+} + 2e^- \rightarrow Sn$$
$$Sn^{4+} + 4e^- \rightarrow Sn$$
the electrode potentials are; $$E^\circ_{Sn^{2+}/Sn} = -0.140$$ V and $$E^\circ_{Sn^{4+}/Sn} = 0.010$$ V. The magnitude of standard electrode potential for $$Sn^{4+}/Sn^{2+}$$ i.e. $$E^\circ_{Sn^{4+}/Sn^{2+}}$$ is ______ $$\times 10^{-2}$$ V (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 56
A radioactive element has a half life of 200 days. The percentage of original activity remaining after 83 days is ______ (Nearest integer)
(Given: antilog 0.125 = 1.333, antilog 0.693 = 4.93)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 57
$$[Fe(CN)_6]^{4-}$$
$$[Fe(CN)_6]^{3-}$$
$$[Ti(CN)_6]^{3-}$$
$$[Ni(CN)_4]^{2-}$$
$$[Co(CN)_6]^{3-}$$
Among the given complexes, number of paramagnetic complexes is
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 58
(a) CoCl$$_3$$ · 4NH$$_3$$,
(b) CoCl$$_3$$ · 5NH$$_3$$,
(c) CoCl$$_3$$ · 6NH$$_3$$ and
(d) CoCl(NO$$_3$$)$$_2$$ · 5NH$$_3$$
Number of complex(es) which will exist in cis-trans form is/are
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 59
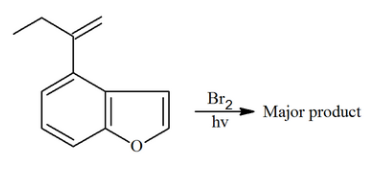
The major product of the following reaction contains ______ bromine atom(s).
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 2 - Chemistry - Question 60
2.5 g of protein containing only glycine (C$$_2$$H$$_5$$NO$$_2$$) is dissolved in water to make 500 mL of solution. The osmotic pressure of this solution at 300 K is found to be $$5.03 \times 10^{-3}$$ bar. The total number of glycine units present in the protein is
(Given: R = 0.083 L bar K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




