NTA JEE Main 28th June 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 28th June 2022 Shift 1 - Question 31
Element "E" belongs to the period 4 and group 16 of the periodic table. The valence shell electron configuration of the element, which is just above "E" in the group is
NTA JEE Main 28th June 2022 Shift 1 - Question 32
Which one of the following techniques is not used to spot components of a mixture separated on thin layer chromatographic plate?
NTA JEE Main 28th June 2022 Shift 1 - Question 33
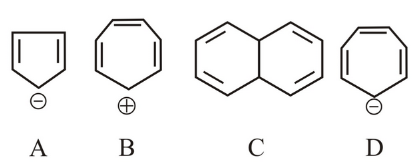
Which of the following structures are aromatic in nature?
NTA JEE Main 28th June 2022 Shift 1 - Question 34
The formula of the purple colour formed in Lassaigne's test for sulphur using sodium nitroprusside is
NTA JEE Main 28th June 2022 Shift 1 - Question 35
Which amongst the following is not a pesticide?
NTA JEE Main 28th June 2022 Shift 1 - Question 36
The incorrect statement about the imperfections in solids is
NTA JEE Main 28th June 2022 Shift 1 - Question 37
The Zeta potential is related to which property of colloids?
NTA JEE Main 28th June 2022 Shift 1 - Question 38
Given are two statements one is labelled as Assertion and other is labelled as Reason.
Assertion: Magnesium can reduce $$Al_2O_3$$ at a temperature below $$1350°$$C, while above $$1350°$$C aluminium can reduce MgO.
Reason: The melting and boiling points of magnesium are lower than those of aluminium.
NTA JEE Main 28th June 2022 Shift 1 - Question 39
Nitrogen gas is obtained by thermal decomposition of:
NTA JEE Main 28th June 2022 Shift 1 - Question 40
Given below are two statements :
Statement I: The pentavalent oxide of group-15 element, $$E_2O_5$$, is less acidic than trivalent oxide, $$E_2O_3$$, of the same element.
Statement II : The acidic character of trivalent oxide of group 15 elements, $$E_2O_3$$, decreases down the group.


