NTA JEE Main 28th June 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 31
Element "E" belongs to the period 4 and group 16 of the periodic table. The valence shell electron configuration of the element, which is just above "E" in the group is
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 32
Which one of the following techniques is not used to spot components of a mixture separated on thin layer chromatographic plate?
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 33
Which of the following structures are aromatic in nature?
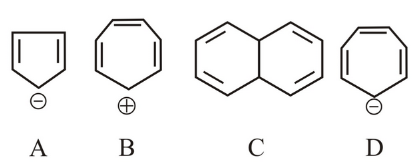
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 34
The formula of the purple colour formed in Lassaigne's test for sulphur using sodium nitroprusside is
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 35
Which amongst the following is not a pesticide?
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 36
The incorrect statement about the imperfections in solids is
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 37
The Zeta potential is related to which property of colloids?
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 38
Given are two statements one is labelled as Assertion and other is labelled as Reason.
Assertion: Magnesium can reduce $$Al_2O_3$$ at a temperature below $$1350°$$C, while above $$1350°$$C aluminium can reduce MgO.
Reason: The melting and boiling points of magnesium are lower than those of aluminium.
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 39
Nitrogen gas is obtained by thermal decomposition of:
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 40
Given below are two statements :
Statement I: The pentavalent oxide of group-15 element, $$E_2O_5$$, is less acidic than trivalent oxide, $$E_2O_3$$, of the same element.
Statement II : The acidic character of trivalent oxide of group 15 elements, $$E_2O_3$$, decreases down the group.
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 41
Dihydrogen reacts with CuO to give
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 42
Which one of the lanthanoids given below is the most stable in divalent form?
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 43
Given below are two statements :
Statement I: $$[Ni(CN)_4]^{2-}$$ is square planar and diamagnetic complex, with $$dsp^2$$ hybridization for Ni but $$[Ni(CO)_4]$$ is tetrahedral, paramagnetic and with $$sp^3$$ hybridication for Ni.
Statement II : $$[NiCl_4]^{2-}$$ and $$[Ni(CO)_4]$$ both have same d-electron configuration, have same geometry and are paramagnetic.
In light the above statements, choose the correct answer form the options given below
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 44
The major product (P) in the reaction
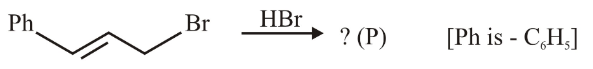
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 45
Which one of the following compounds is inactive towards $$S_N1$$ reaction?
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 46
The correct structure of product 'A' formed in the following reaction,
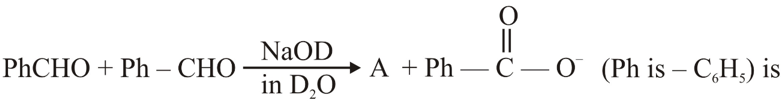
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 47
Identify the major product formed in the following sequence of reactions :
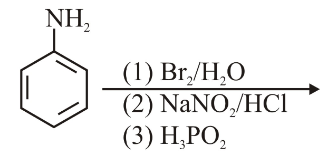
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 48
A primary aliphatic amine on reaction with nitrous acid in cold (273 K) and there after raising temperature of reaction mixture to room temperature (298 K), gives a/an
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 49
Which one of the following is NOT a copolymer?
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 50
Stability of $$\alpha$$-Helix structure of proteins depends upon
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 51
If the work function of a metal is $$6.63 \times 10^{-19}$$ J, the maximum wavelength of the photon required to remove a photoelectron from the metal is ______ nm. Nearest integer
[Given : $$h = 6.63 \times 10^{-34}$$ J s, and $$c = 3 \times 10^{8}$$ m s$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 52
The hybridization of P exhibited in $$PF_5$$ is $$sp^x d^y$$. The value of $$y$$ is ______
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 53
$$4.0$$ L of an ideal gas is allowed to expand isothermally into vacuum until the total volume is $$20$$ L. The amount of heat absorbed in this expansion is ______ L atm.
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 54
A $$2.0$$ g sample containing $$MnO_2$$ is treated with HCl liberating $$Cl_2$$. The $$Cl_2$$ gas is passed into a solution of KI and $$60.0$$ mL of $$0.1$$ M $$Na_2S_2O_3$$ is required to titrate the liberated iodine. The percentage of $$MnO_2$$ in the sample is ______ Nearest integer
[Atomic masses (in u) Mn = 55; Cl = 35.5; O = 16, I = 127, Na = 23, K = 39, S = 32]
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 55
In the estimation of bromine, $$0.5$$ g of an organic compound gave $$0.40$$ g of silver bromide. The percentage of bromine in the given compound is ______ % (nearest integer)
(Relative atomic masses of Ag and Br are 108u and 80u, respectively).
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 56
The vapour pressures of two volatile liquids A and B at $$25°$$C are $$50$$ Torr and $$100$$ Torr, respectively. If the liquid mixture contains $$0.3$$ mole fraction of A, then the mole fraction of liquid B in the vapour phase is $$\frac{x}{17}$$. The value of $$x$$ is ______
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 57
The solubility product of a sparingly soluble salt $$A_2X_3$$ is $$1.1 \times 10^{-23}$$. If specific conductance of the solution is $$x \times 10^{-3}$$ S m$$^2$$ mol$$^{-1}$$. The value of $$x$$ is ______
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 58
The quantity of electricity in Faraday needed to reduce $$1$$ mol of $$Cr_2O_7^{2-}$$ to $$Cr^{3+}$$ is ______
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 59
For a first order reaction A $$\to$$ B, the rate constant, $$k = 5.5 \times 10^{-14}$$ s$$^{-1}$$. The time required for $$67\%$$ completion of reaction is $$x \times 10^{-1}$$ times the half life of reaction. The value of $$x$$ is ______ Nearest integer) (Given : $$\log 3 = 0.4771$$)
789
456
123
0.-
Clear All
NTA JEE Main 28th June 2022 Shift 1 - Chemistry - Question 60
Number of complexes which will exhibit synergic bonding amongst, $$[Cr(CO)_6], [Mn(CO)_5]$$ and $$[Mn_2(CO)_{10}]$$ is ______
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




