NTA JEE Main 28th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 28th July 2022 Shift 2 - Question 51
2 L of 0.2 M $$H_2SO_4$$ is reacted with 2 L of 0.1 M NaOH solution, the molarity of the resulting product $$Na_2SO_4$$ in the solution is _____ millimolar.
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 52
If the wavelength for an electron emitted from $$H^-$$ atom is $$3.3 \times 10^{-10}$$ m, then energy absorbed by the electron in its ground state compared to minimum energy required for its escape from the atom, is _____ times. [Given: $$h = 6.626 \times 10^{-34}$$ Js, Mass of electron $$= 9.1 \times 10^{-31}$$ kg]
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 53
Among the following the number of state variable is
Internal energy (U), Volume (V), Heat (q), Enthalpy (H)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 54
At 600 K, 2 mol of NO are mixed with 1 mol of $$O_2$$.
$$2NO(g) + O_2(g) \rightleftharpoons 2NO_2(g)$$
The reaction occurring as comes to equilibrium under a total pressure of 1 atm. Analysis of the system shows that 0.6 mol of oxygen are present at equilibrium. The equilibrium constant for the reaction is _____
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 55
On reaction with stronger oxidizing agent like $$KIO_4$$, hydrogen peroxide oxidizes with the evolution of $$O_2$$. The oxidation number of I in $$KIO_4$$ changes to
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 56
A sample of 0.125 g of an organic compound when analysed by Duma's method yields 22.78 mL of nitrogen gas collected over KOH solution at 280 K and 759 mmHg. The percentage of nitrogen in the given organic compound is _____
(a) The vapour pressure of water at 280 K is 14.2 mmHg
(b) $$R = 0.082$$ L atm K$$^{-1}$$ mol$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 57
Metal M crystallizes into a FCC lattice with the edge length of $$4.0 \times 10^{-8}$$ cm. The atomic mass of the metal is _____ g/mol. (Use: $$N_A = 6.02 \times 10^{23}$$ mol$$^{-1}$$, density of metal, $$M = 9.03$$ g cm$$^{-3}$$)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 58
A gaseous mixture of two substances A and B, under a total pressure of 0.8 atm is in equilibrium with an ideal liquid solution. The mole fraction of substance A is 0.5 in the vapour phase and 0.2 in the liquid phase. The vapour pressure of pure liquid A is _____ atm. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 28th July 2022 Shift 2 - Question 59
For a reaction, given below is the graph of $$\ln k$$ vs $$\frac{1}{T}$$. The activation energy for the reaction is equal to _____ cal mol$$^{-1}$$. (Given: $$R = 2$$ cal K$$^{-1}$$ mol$$^{-1}$$)
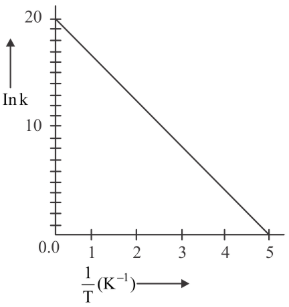
789
456
123
0.-
Clear All
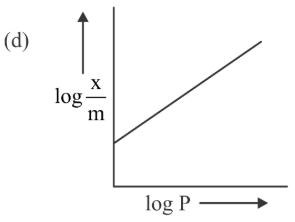
NTA JEE Main 28th July 2022 Shift 2 - Question 60
Among the following the number of curves not in accordance with Freundlich adsorption isotherm is
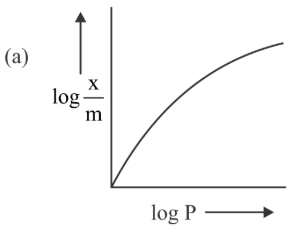
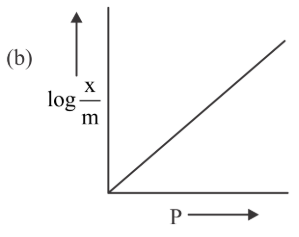
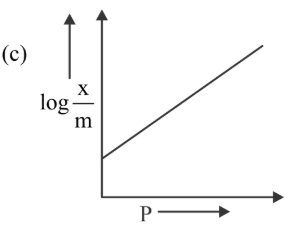
789
456
123
0.-
Clear All


