NTA JEE Main 28th July 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 28th July 2022 Shift 1 - Question 41
Given below are two statements:
Statement I: In polluted water values of both dissolved oxygen and BOD are very low.
Statement II: Eutrophication results in decrease in the amount of dissolved oxygen.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 28th July 2022 Shift 1 - Question 42
Match List-I with List-II.
| List-I | List-II |
|---|---|
| A. $$Cd(s) + 2Ni(OH)_3(s) \to CdO(s) + 2Ni(OH)_2(s) + H_2O(l)$$ | I. Primary battery |
| B. $$Zn(Hg) + HgO(s) \to ZnO(s) + Hg(l)$$ | II. Discharging of secondary battery |
| C. $$2PbSO_4(s) + 2H_2O(l) \to Pb(s) + PbO_2(s) + 2H_2SO_4(aq)$$ | III. Fuel cell |
| D. $$2H_2(g) + O_2(g) \to 2H_2O(l)$$ | IV. Charging of secondary battery |
NTA JEE Main 28th July 2022 Shift 1 - Question 43
Which of the reaction is suitable for concentrating ore by leaching process?
NTA JEE Main 28th July 2022 Shift 1 - Question 44
Match List-I with List-II.
| List-I | List-II |
|---|---|
| A. $$4NH_3(g) + 5O_2(g) \to 4NO(g) + 6H_2O(g)$$ | I. NO(g) |
| B. $$N_2(g) + 3H_2(g) \to 2NH_3(g)$$ | II. $$H_2SO_4(l)$$ |
| C. $$C_{12}H_{22}O_{11}(aq) + H_2O(l) \to C_6H_{12}O_6 (Glucose) + C_6H_{12}O_6 (Fructose)$$ | III. Pt(s) |
| D. $$2SO_2(g) + O_2(g) \to 2SO_3(g)$$ | IV. Fe(s) |
NTA JEE Main 28th July 2022 Shift 1 - Question 45
Match List-I with List-II, match the gas evolved during each reaction.
| List-I | List-II |
|---|---|
| A. $$(NH_4)_2Cr_2O_7 \xrightarrow{\Delta}$$ | I. $$H_2$$ |
| B. $$KMnO_4 + HCl \to$$ | II. $$N_2$$ |
| C. $$Al + NaOH + H_2O \to$$ | III. $$O_2$$ |
| D. $$NaNO_3 \xrightarrow{\Delta}$$ | IV. $$Cl_2$$ |
Choose the correct answer from the options given below
NTA JEE Main 28th July 2022 Shift 1 - Question 46
Which of the following has least tendency to liberate $$H_2$$ from mineral acids?
NTA JEE Main 28th July 2022 Shift 1 - Question 47
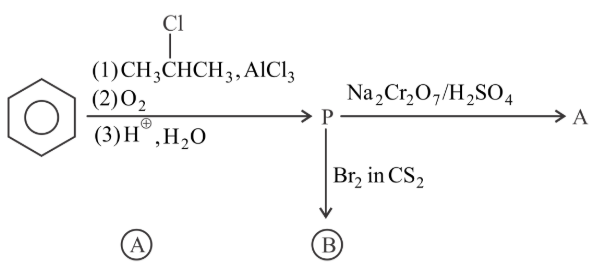
Identify the major product A and B for the below given reaction sequence.
NTA JEE Main 28th July 2022 Shift 1 - Question 48
Terylene polymer is obtained by condensation of
NTA JEE Main 28th July 2022 Shift 1 - Question 49
Statements about Enzyme Inhibitor Drugs are given below:
(A) There are Competitive and Non-competitive inhibitor drugs.
(B) These can bind at the active sites and allosteric sites.
(C) Competitive Drugs are allosteric site blocking drugs.
(D) Non-competitive Drugs are active site blocking drugs.
Choose the correct answer from the options given below:
NTA JEE Main 28th July 2022 Shift 1 - Question 50
For the below given cyclic hemiacetal X, the correct pyranose structure is