NTA JEE Main 27th June 2022 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 31
The correct order of ionic size of $$N^{3-}, Na^+, F^-, Mg^{2+}$$ and $$O^{2-}$$ is :
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 32
Identify the incorrect statement for $$PCl_5$$ from the following.
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 33
The correct order of increasing intermolecular hydrogen bond strength is
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 34
Which amongst the given plots is the correct plot for pressure (p) vs density (d) for an ideal gas?
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 35
Which of the following is most stable?
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 36
What will be the major product of following sequence of reactions?
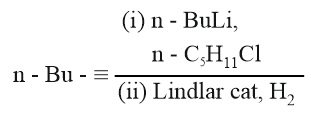
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 37
Product 'A' of following sequence of reactions is
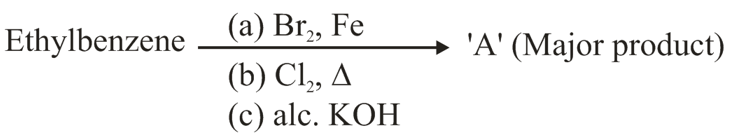
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 38
In 3d series, the metal having the highest $$M^{2+}/M$$ standard electrode potential is
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 39
Statement I: Leaching of gold with cyanide ion in absence of air/$$O_2$$ leads to cyano complex of Au(III).
Statement II: Zinc is oxidized during the displacement reaction carried out for gold extraction.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 40
The gas produced by treating an aqueous solution of ammonium chloride with sodium nitrite is
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 41
Given below are two statements: one is labelled as Assertion and the other is labelled as Reason.
Assertion: Flourine forms one oxoacid.
Reason: Flourine has smallest size amongst all halogens and is highly electronegative.
In the light of the above statements, choose the most appropriate answer from the Question: options given
below.
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 42
On the surface of polar stratospheric clouds, hydrolysis of chlorine nitrate gives A and B while its reaction with HCl produces B and C. A, B and C are, respectively
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 43
The 'f' orbitals are half and completely filled, respectively in lanthanide ions [Given : Atomic no. Eu, 63 : Sm, 62 : Tm, 69; Tb, 65; Yb, 70; Dy, 66]
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 44
Arrange the following coordination compounds in the increasing order of magnetic moments. (Atomic numbers: Mn = 25; Fe = 26)
A. $$[FeF_6]^{3-}$$
B. $$[Fe(CN)_6]^{3-}$$
C. $$[MnCl_6]^{3-}$$ (high spin)
D. $$[Mn(CN)_6]^{3-}$$
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 45
Match List-I with List-II
List - I List - II
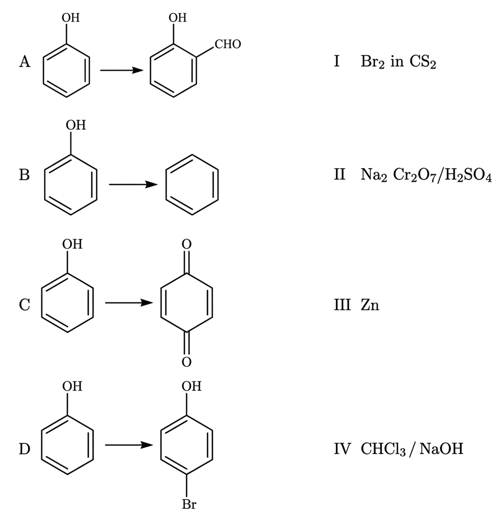
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 46
Decarboxylation of all six possible forms of diaminobenzoic acids $$C_6H_3(NH_2)_2COOH$$ yields three products A, B and C. Three acids give a product 'A', two acids give a product 'B' and one acid give a product 'C'. The melting point of product 'C' is
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 47
Which is true about Buna-N?
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 48
Match List - I with List - II.
List - I List - II
| (A) Antipyretic | (I) Reduces pain |
| (B) Analgesic | (II) Reduces stress |
| (C) Tranquilizer | (III) Reduces fever |
| (D) Antacid | (IV) Reduces acidity(stomach) |
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 49
Match List - I with List - II.
| List-I (Anion) | List-II (gas evolved on reaction with dil. $$H_2SO_4$$) |
|---|---|
| (A) $$CO_3^{2-}$$ | (I) Colourless gas which turns lead acetate paper black. |
| (B) $$S^{2-}$$ | (II) Colourless gas which turns acidified potassium dichromate solution green. |
| (C) $$SO_3^{2-}$$ | (III) Brown fumes which turns acidified KI solution containing starch blue. |
| (D) $$NO_2^-$$ | (IV) Colourless gas evolved with brisk effervescence, which turns lime water milky. |
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 50
Given below are two statements.
Statement I: Maltose has two $$\alpha - D$$ - glucose units linked at $$C_1$$ and $$C_4$$ and is a reducing sugar.
Statement II: Maltose has two monosaccharides: $$\alpha$$-D-glucose and $$\beta$$-D-glucose linked at $$C_1$$ and $$C_6$$ and it is a non-reducing sugar.
In the light of the above statements, choose the correct answer from the options Question: given below.
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 51
$$116$$ g of a substance upon dissociation reaction, yields $$7.5$$ g of hydrogen, $$60$$ g of oxygen and $$48.5$$ g of carbon. Given that the atomic masses of H, O and C are 1, 16 and 12, respectively. The data agrees with how many formulae of the following?
A. $$CH_3COOH$$
B. HCHO
C. $$CH_3OOCH_3$$
D. $$CH_3CHO$$
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 52
Consider the following set of quantum numbers.
A: n=3, l=3, m$$_{1}$$=-3
B: n=3, l=2, m$$_{1}$$=-2
C: n=2, l=1, m$$_{1}$$=+1
D: n=2, l=2, m$$_{1}$$=+2
The number of correct sets of quantum numbers is ______
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 53
When 5 moles of He gas expand isothermally and reversibly at $$300$$ K from $$10$$ litre to $$20$$ litre, the magnitude of the maximum work obtained is ______ J. [nearest integer] (Given : $$R = 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$ and $$\log 2 = 0.3010$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 54
pH value of $$0.001$$ M NaOH solution is ______
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 55
BeO reacts with HF in presence of ammonia to give [A] which on thermal decomposition produces [B] and ammonium fluoride. Oxidation state of Be in [A] is ______
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 56
$$0.25$$ g of an organic compound containing chlorine gave $$0.40$$ g of silver chloride in Carius estimation. The percentage of chlorine present in the compound is ______ [in nearest integer]
(Given: Molar mass of Ag is $$108$$ g mol$$^{-1}$$ and that of Cl is $$35.5$$ g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 57
A solution containing $$2.5 \times 10^{-3}$$ kg of a solute dissolved in $$75 \times 10^{-3}$$ kg of water boils at $$373.535$$ K. The molar mass of the solute is ______ mol$$^{-1}$$. [nearest integer] (Given : $$K_b(H_2O) = 0.52$$ K kg mol$$^{-1}$$ and boiling point of water $$= 373.15$$ K)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 58
For the reaction taking place in the cell:
$$Pt(s)|H_2(g)|H^+(aq) || Ag^+(aq)|Ag(s)$$
$$E_{cell} = +0.5332$$ V.
The value of $$\Delta_r G^\circ$$ is ______ kJ mol$$^{-1}$$. (in nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 59
It has been found that for a chemical reaction with rise in temperature by $$9$$ K the rate constant gets doubled. Assuming a reaction to be occurring at $$300$$ K, the value of activation energy is found to be ______ kJ mol$$^{-1}$$. [nearest integer] (Given $$\ln 10 = 2.3, R = 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$, $$\log 2 = 0.30$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 2 - Chemistry - Question 60
A Freundlich adsorption isotherm graph is shown.
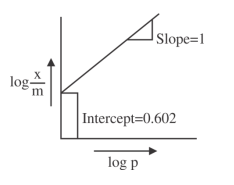
If the initial pressure of a gas is $$0.03$$ atm, the mass of the gas adsorbed per gram of the adsorbent is ______ $$\times 10^{-2}$$ g
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




