NTA JEE Main 27th June 2022 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 31
Given below are two statements : one is labelled as Assertion and the other is labelled as Reason.
Assertion: The ionic radii of $$O^{2-}$$ and $$Mg^{2+}$$ are same.
Reason: Both $$O^{2-}$$ and $$Mg^{2+}$$ are isoelectronic species.
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 32
Based upon VSEPR theory, match the shape (geometry) of the molecules in List-I with the molecules in List-II
| List-I (Shape) | List-II (Molecules) |
|---|---|
| A T-shaped | I. $$XeF_4$$ |
| B Trigonal planar | II. $$SF_4$$ |
| C Square planar | III. $$ClF_3$$ |
| D See-saw | IV. $$BF_3$$ |
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 33
Match List - I with List - II.
| List-I | List-II |
|---|---|
| (A) Spontaneous process | (I) $$\Delta H < 0$$ |
| (B) Process with $$\Delta P = 0, \Delta T = 0$$ | (II) $$\Delta G_{T,P} < 0$$ |
| (C) $$\Delta H_{reaction}$$ | (III) Isothermal and isobaric process |
| (D) Exothermic Process | (IV) [Bond energies in reactants] - [Bond energies in products] |
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 34
Addition of $$H_2SO_4$$ to $$BaO_2$$ produces
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 35
$$BeCl_2$$ reacts with $$LiAlH_4$$ to give
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 36
Match List - I with List - II.
| List-I (Si-Compounds) | List-II (Si-Polymeric/Other Products) |
|---|---|
| A $$(CH_3)_4Si$$ | I Chain Silicone |
| B $$(CH_3)Si(OH)_3$$ | II Dimeric Silicone |
| C $$(CH_3)_2Si(OH)_2$$ | III Silane |
| D $$(CH_3)_3Si(OH)$$ | IV 2D-Silicone |
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 37
L-isomer of a compound 'A'($$C_4H_8O_4$$) gives a positive test with $$[Ag(NH_3)_2]^+$$. Treatment of 'A' with acetic anhydride yields triacetate derivative. Compound 'A' produces an optically active compound (B) and an optically inactive compound (C) on treatment with bromine water and $$HNO_3$$ respectively. Compound (A) is :
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 38
Given below are two Statements :
Statement I : Classical smog occurs in cool humid climate. It is a reducing mixture of smoke, fog and sulphur dioxide.
Statement II : Photochemical smog has components, ozone, nitric oxide, acrolein, formaldehyde, PAN etc.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 39
Given below are two statements : one is labelled as Assertion and the other is labelled as Reason.
Assertion: At $$10°$$C, the density of a $$5$$ M solution of KCl [atomic masses of K & Cl are $$39$$ & $$35.5$$ g mol$$^{-1}$$ respectively], is 'x' g mL$$^{-1}$$. The solution is cooled to $$-21°$$C. The molality of the solution will remain unchanged.
Reason: The molality of a solution does not change with temperature as mass remains unaffected with temperature.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 40
Match List - I with List - II.
| List-I | List-II |
|---|---|
| A Lyophilic colloid | I Liquid-liquid colloid |
| B Emulsion | II Protective colloid |
| C Positively charged colloid | III $$FeCl_3 + NaOH$$ |
| D Negatively charged colloid | IV $$FeCl_3$$ + hot water |
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 41
Match List - I with List - II.
| List-I | List-II |
|---|---|
| A Concentration of gold ore | I Aniline |
| B Leaching of alumina | II NaOH |
| C Froth stabiliser | III $$SO_2$$ |
| D Blister copper | IV NaCN |
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 42
Heating white phosphorus with conc. NaOH solution gives mainly
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 43
Which of the following will have maximum stabilization due to crystal field?
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 44
The major product of the following reaction is
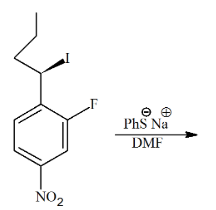
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 45
Which of the following reactions will yield benzaldehyde as a product?
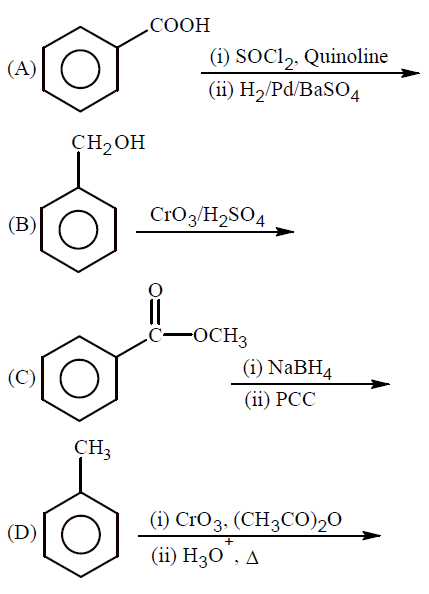
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 46
'A' and 'B' respectively are
A $$\xrightarrow[(2) Zn-H_2O]{(1) O_3 \atop}$$ Ethane-1,2-dicarbaldehyde + Glyoxal/Oxaldehyde
B $$\xrightarrow[(2) Zn-H_2O]{(1) O_3 \atop}$$ 5-oxohexanal
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 47
Given below are two statements :
Statement - I : In Hofmann degradation reaction, the migration of only an alkyl group takes place from carbonyl carbon of the amide to the nitrogen atom.
Statement - II : The group is migrated in Hofmann degradation reaction to electron deficient atom.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 48
Match List - I with List - II.
| List-I (Polymer) | List-II (Used in) |
|---|---|
| A Bakelite | I Radio and television cabinets |
| B Glyptal | II Electrical switches |
| C PVC | III Paints and Lacquers |
| D Polystyrene | IV Water pipes |
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 49
Match List - I with List - II.
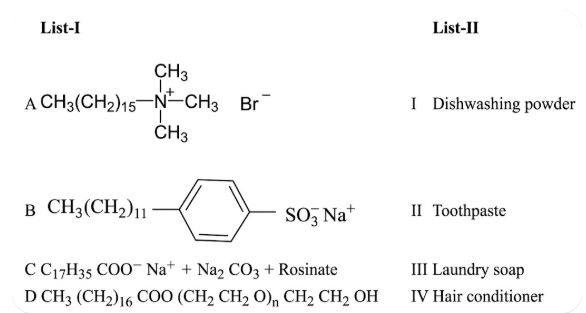
Choose the correct answer from the options given below
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 50
Which of the following is structure of a separating funnel?
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 51
If the uncertainty in velocity and position of a minute particle in space are, $$2.4 \times 10^{-26}$$ (ms$$^{-1}$$) and $$10^{-7}$$ (m) respectively. The mass of the particle in g is ______ (Nearest integer)
(Given : $$h = 6.626 \times 10^{-34}$$ Js)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 52
$$2NOCl(g) \rightleftharpoons 2NO(g) + Cl_2(g)$$
In an experiment, $$2.0$$ moles of NOCl was placed in a one-litre flask and the concentration of NO after equilibrium established, was found to be $$0.4$$ mol/L. The equilibrium constant at $$30°$$C is ______ $$\times 10^{-4}$$.
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 53
Total number of possible stereoisomers of dimethyl cyclopentane is ______
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 54
Metal deficiency defect is shown by $$Fe_{0.93}O$$. In the crystal, some $$Fe^{2+}$$ cations are missing and loss of positive charge is compensated by the presence of $$Fe^{3+}$$ ions. The percentage of $$Fe^{2+}$$ ions in the $$Fe_{0.93}O$$ crystals is ______ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 55
Two elements A and B which form $$0.15$$ moles of $$A_2B$$ and $$AB_3$$ type compounds. If both $$A_2B$$ and $$AB_3$$ weigh equally, then the atomic weight of A is ______ times of atomic weight of B.
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 56
$$2$$ g of a non-volatile non-electrolyte solute is dissolved in $$200$$ g of two different solvents A and B whose ebullioscopic constants are in the ratio of $$1 : 8$$. The elevation in boiling points of A and B are in the ratio $$\frac{x}{y}$$ ($$x : y$$). The value of $$y$$ is ______ (Nearest Integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 57
The limiting molar conductivities of NaI, $$NaNO_3$$ and $$AgNO_3$$ are $$12.7, 12.0$$ and $$13.3$$ mS m$$^2$$ mol$$^{-1}$$, respectively (all at $$25°$$C). The limiting molar conductivity of AgI at this temperature is ______ mS m$$^2$$ mol$$^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 58
The rate constant for a first order reaction is given by the following equation :
$$\ln k = 33.24 - \frac{2.0 \times 10^4 K}{T}$$
The Activation energy for the reaction is given by ______ kJ mol$$^{-1}$$. (In Nearest integer) (Given : $$R = 8.3$$ J K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 59
The number of statement(s) correct from the following for Copper is/are
(A) Cu(II) complexes are always paramagnetic
(B) Cu(I) complexes are generally colourless
(C) Cu(I) is easily oxidized
(D) In Fehling solution, the active reagent has Cu(I)
789
456
123
0.-
Clear All
NTA JEE Main 27th June 2022 Shift 1 - Chemistry - Question 60
Acidified potassium permanganate solution oxidises oxalic acid. The spin-only magnetic moment of the mangenese product formed from the above reaction is ______ B.M. (Nearest Integer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




