NTA JEE Main 27th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 27th July 2022 Shift 2 - Question 51
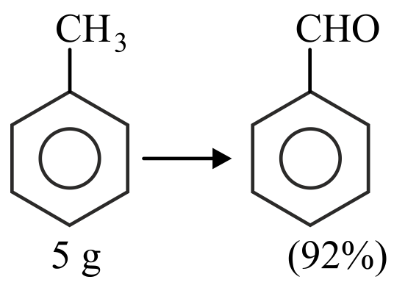
In the above reaction,5 g of toluene is converted into benzaldehyde with 92% yield. The amount of benzaldehyde produced is _____ $$\times 10^{-2}$$ g
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 52
The number of molecule(s) or ion(s) from the following having non-planar structure is ______.
$$NO_3^-$$, $$H_2O_2$$, $$BF_3$$, $$PCl_3$$, $$XeF_4$$, $$SF_4$$, $$XeO_3$$, $$PH_4^+$$, $$SO_3$$, $$[Al(OH)_4]^-$$
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 53
For a real gas at $$25^\circ C$$ temperature and high pressure (99 bar) the value of compressibility factor is 2, so the value of Van der Waal's constant 'b' should be _____ $$\times 10^{-2}$$ L mol$$^{-1}$$. (Given $$R = 0.083$$ L bar K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 54
A gas (Molar mass = 280 g mol$$^{-1}$$) was burnt in excess $$O_2$$ in a constant volume calorimeter and during combustion the temperature of calorimeter increased from 298.0 K to 298.45 K. If the heat capacity of calorimeter is 2.5 kJ K$$^{-1}$$ and enthalpy of combustion of gas is 9 kJ mol$$^{-1}$$ then amount of gas burnt is _____ g.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 55
The normality of $$H_2SO_4$$ in the solution obtained on mixing 100 mL of 0.1 M $$H_2SO_4$$ with 50 mL of 0.1 M NaOH is _____ $$\times 10^{-1}$$ N.
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 56
When a certain amount of solid A is dissolved in 100 g of water at $$25^\circ C$$ to make a dilute solution, the vapour pressure of the solution is reduced to one-half of that of pure water. The vapour pressure of pure water is 23.76 mmHg. The number of moles of solute A added is
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 57
$$[A] \xrightarrow{} [B]$$
Reactant $$\to$$ Product
If formation of compound [B] follows the first order of kinetics and after 70 minutes the concentration of [A] was found to be half of its initial concentration. Then the rate constant of the reaction is $$x \times 10^{-6} s^{-1}$$. The value of $$x$$ is ____
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 58
Among the following ores Bauxite, Siderite, Cuprite, Calamine, Haematite, Kaolinite, Malachite, Magnetite, Sphalerite, Limonite, Cryolite, the number of principal ores of iron is
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 59
The oxidation state of manganese in the product obtained in a reaction of potassium permanganate and hydrogen peroxide in basic medium is
789
456
123
0.-
Clear All
NTA JEE Main 27th July 2022 Shift 2 - Question 60
The spin only magnetic moment of the complex present in Fehling's reagent is _____ B.M. (Round off your answer to the nearest integer)
789
456
123
0.-
Clear All


