NTA JEE Main 27th July 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 27th July 2021 Shift 2 - Question 31
If the Thomson model of the atom was correct, then the result of Rutherford's gold foil experiment would have been:
NTA JEE Main 27th July 2021 Shift 2 - Question 32
The CORRECT order of first ionisation enthalpy is:
NTA JEE Main 27th July 2021 Shift 2 - Question 33
The number of neutrons and electrons, respectively, present in the radioactive isotope of hydrogen is:
NTA JEE Main 27th July 2021 Shift 2 - Question 34
Match List - I with List II:
List-I List-II
a. Li i. photoelectric cell
b. Na ii. absorbent of CO$$_2$$
c. K iii. coolant in fast breeder nuclear reactor
d. Cs iv. treatment of cancer
v. bearings for motor engines
Choose the correct answer from the options given below:
NTA JEE Main 27th July 2021 Shift 2 - Question 35
Which one of the following set of elements can be detected using sodium fusion extract?
NTA JEE Main 27th July 2021 Shift 2 - Question 36
Given below are two statements:
Statement I : Hyperconjugation is a permanent effect.
Statement II : Hyperconjugation in ethyl cation $$\left(CH_3 - \overset{+}{CH_2}\right)$$ involves the overlapping of $$C_{sp^2} - H_{1s}$$ bond with empty 2p orbital of other carbon.
Choose the correct option:
NTA JEE Main 27th July 2021 Shift 2 - Question 37
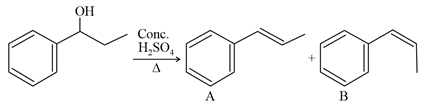
Consider the above reaction, and choose the correct statement:
NTA JEE Main 27th July 2021 Shift 2 - Question 38
The correct sequence of correct reagents for the following transformation is:-
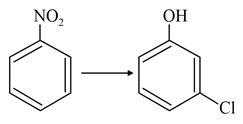
NTA JEE Main 27th July 2021 Shift 2 - Question 39
Match List - I with List - II:
List - I (compound) List - II (effect/affected species)
a. Carbon monoxide i. Carcinogenic
b. Sulphur dioxide ii. Metabolized by pyrus plants
c. Polychlorinated biphenyls iii. Haemoglobin
d. Oxides of Nitrogen iv. Stiffness of flower buds
Choose the correct answer from the options given below:
NTA JEE Main 27th July 2021 Shift 2 - Question 40
Select the correct statements.
(A) Crystalline solids have long range order.
(B) Crystalline solids are isotropic.
(C) Amorphous solid are sometimes called pseudo solids.
(D) Amorphous solids soften over a range of temperatures.
(E) Amorphous solids have a definite heat of fusion.
Choose the most appropriate answer from the options given below.


