NTA JEE Main 27th August 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 27th August 2021 Shift 2 - Question 51
100 g of propane is completely reacted with 1000 g of oxygen. The mole fraction of carbon dioxide in the resulting mixture is $$x \times 10^{-2}$$. The value of $$x$$ is _________. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 52
The number of photons emitted by a monochromatic (single frequency) infrared range finder of power 1 mW and wavelength of 1000 nm, in 0.1 second is $$x \times 10^{13}$$. The value of x is _________. (Nearest integer)
(h = $$6.63 \times 10^{-34}$$ Js, c = $$3.00 \times 10^8$$ ms$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 53
The number of species having non-pyramidal shape among the following is:
(A) SO$$_3$$
(B) NO$$_3^-$$
(C) PCl$$_3$$
(D) CO$$_3^{2-}$$
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 54
Two flasks I and II shown below are connected by a valve of negligible volume.
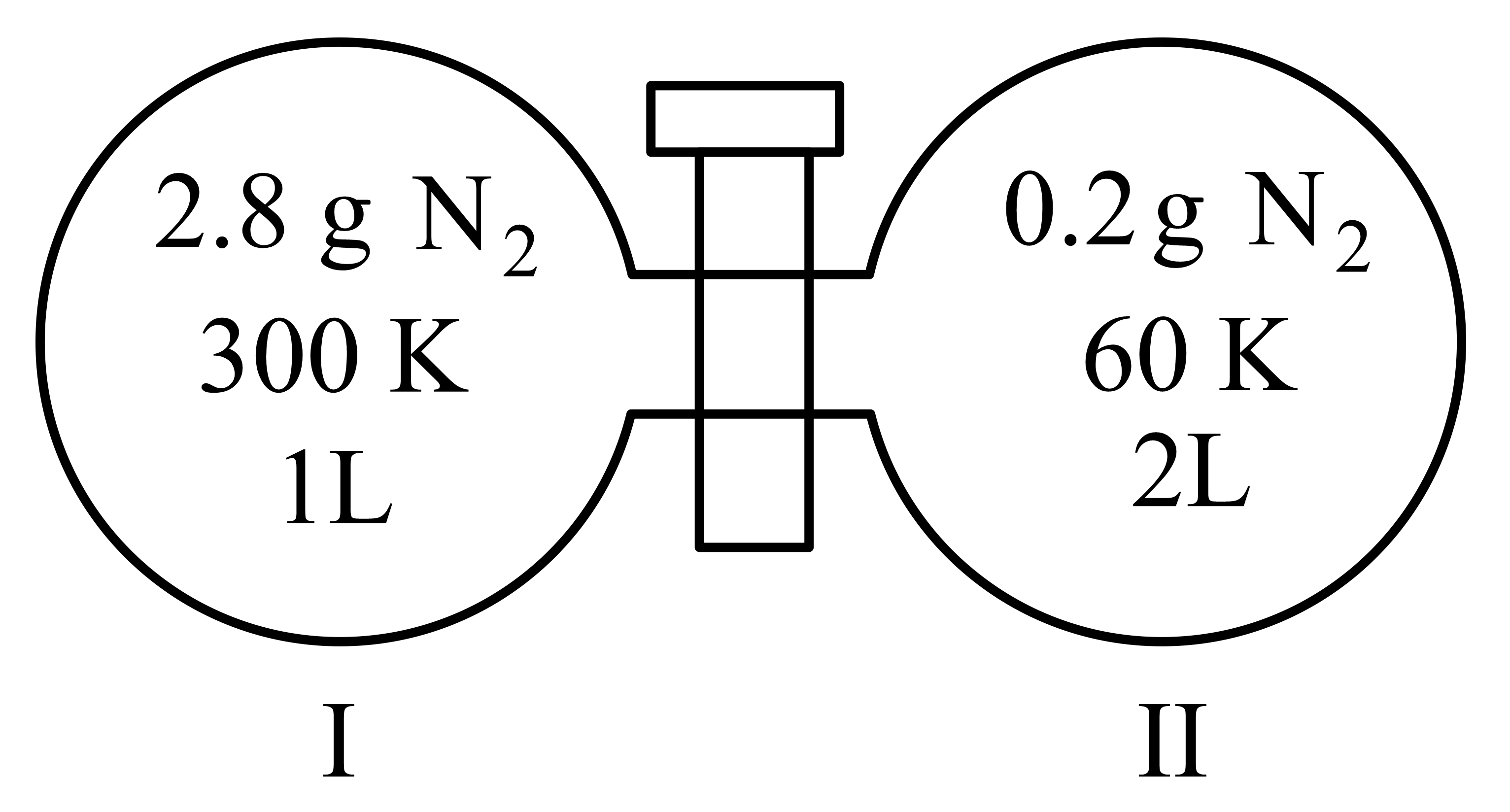
When the valve is opened, the final pressure of the system in bar is $$x \times 10^{-2}$$. The value of x is _________. (Integer answer)
[Assume : Ideal gas; 1 bar = $$10^5$$ Pa; Molar mass of N$$_2$$ = 28.0 mol$$^{-1}$$; R = 8.31 J mol$$^{-1}$$ K$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 55
Data given for the following reaction is as follows:
FeO$$_{(s)}$$ + C$$_{(graphite)}$$ $$\rightarrow$$ Fe$$_{(s)}$$ + CO$$_{(g)}$$
Substance $$\Delta_f H°$$(kJ mol$$^{-1}$$) $$\Delta S°$$(J mol$$^{-1}$$ K$$^{-1}$$)
FeO$$(s)$$ -266.3 57.49
C$$(graphite)$$ 0 5.74
Fe$$(s)$$ 0 27.28
CO$$(g)$$ -110.5 197.6
The minimum temperature in K at which the reaction becomes spontaneous is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 56
When 5.1 g of solid NH$$_4$$HS is introduced into a two litre evacuated flask at 27°C, 20% of the solid decomposes into gaseous ammonia and hydrogen sulphide. The K$$_p$$ for the reaction at 27°C is $$x \times 10^{-2}$$. The value of x is _________. (Integer answer)
[Given 1R = 0.082 L atm K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 57
40 g of glucose (Molar mass = 180) is mixed with 200 mL of water. The freezing point of solution is _________ K. (Nearest integer)
[Given : K$$_f$$ = 1.86 K kg mol$$^{-1}$$; Density of water = 1.00 g cm$$^{-3}$$; Freezing point of water = 273.15 K]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 58
The resistance of conductivity cell with cell constant 1.14 cm$$^{-1}$$, containing 0.001M KCl at 298 K is 1500$$\Omega$$. The molar conductivity of 0.001M KCl solution at 298 K in S cm$$^2$$ mol$$^{-1}$$ is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 59
The first order rate constant for the decomposition of CaCO$$_3$$ at 700 K is $$6.36 \times 10^{-3}$$ s$$^{-1}$$ and activation energy is 209 kJ mol$$^{-1}$$. Its rate constant (in s$$^{-1}$$) at 600 K is $$x \times 10^{-6}$$. The value of x is _________. (Nearest integer)
[Given R = 8.31 J K$$^{-1}$$ mol$$^{-1}$$; log $$6.36 \times 10^{-3}$$ = -2.19, $$10^{-4.79}$$ = $$1.62 \times 10^{-5}$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 2 - Question 60
The number of optical isomers possible for [Cr(C$$_2$$O$$_4$$)$$_3$$]$$^{3-}$$ is:
789
456
123
0.-
Clear All


.webp)
