NTA JEE Main 27th August 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 31
The unit of the van der Waals gas equation parameter 'a' in $$\left(P + \frac{an^2}{V^2}\right)(V - nb) = nRT$$ is:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 32
In polythionic acid, H$$_2$$S$$_x$$O$$_6$$ ($$x$$ = 3 to 5) the oxidation state(s) of sulphur is/are:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 33
Deuterium resembles hydrogen in properties but:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 34
The number of water molecules in gypsum, dead burnt plaster and plaster of Paris, respectively are:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 35
In which one of the following molecules strongest back donation of an electron pair from halide to boron is expected?
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 36
The gas 'A' is having very low reactivity reaches to stratosphere. It is non-toxic and non-flammable but dissociated by UV-radiations in stratosphere. The intermediates formed initially from the gas 'A' are:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 37
Match List - I with List - II:
List-I (Property) List-II (Example)
(a) Diamagnetism (i) MnO
(b) Ferrimagnetism (ii) O$$_2$$
(c) Paramagnetism (iii) NaCl
(d) Antiferromagnetism (iv) Fe$$_3$$O$$_4$$
Choose the most appropriate answer from the options given below:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 38
Tyndall effect is more effectively shown by:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 39
Which refining process is generally used in the purification of low melting metals?
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 40
Match List - I with List - II:
List-I (Species) List-II (Number of lone pairs of electrons on the central atom)
(a) XeF$$_2$$ (i) 0
(b) XeO$$_2$$F$$_2$$ (ii) 1
(c) XeO$$_3$$F$$_2$$ (iii) 2
(d) XeF$$_4$$ (iv) 3
Choose the most appropriate answer from the options given below:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 41
The nature of oxides V$$_2$$O$$_3$$ and CrO is indexed as 'X' and 'Y' type respectively. The correct set of X and Y is:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 42
Acidic ferric chloride solution on treatment with excess of potassium ferrocyanide gives a Prussian blue coloured colloidal species. It is:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 43
In the following sequence of reactions the P is:
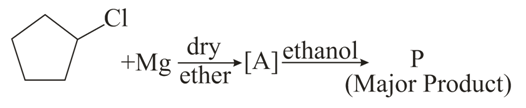
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 44
Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Synthesis of ethyl phenyl ether may be achieved by Williamson synthesis.
Reason (R): Reaction of bromobenzene with sodium ethoxide yields ethyl phenyl ether.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 45
The structure of the starting compound P used in the reaction given below is:
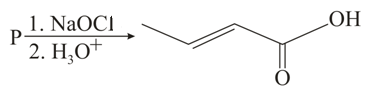
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 46
The major product of the following reaction is:
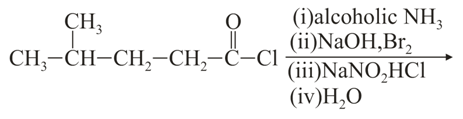
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 47
In the following sequence of reactions, the final product D is:
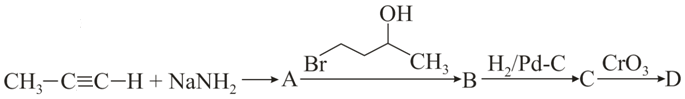
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 48
Which of the following is not a correct statement for primary aliphatic amines?
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 49
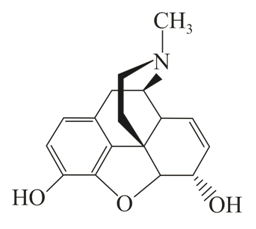
A
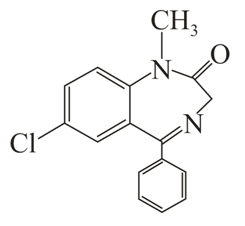
B
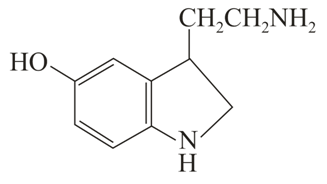
C
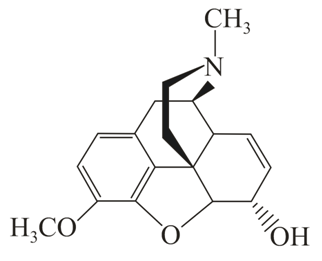
D
The correct statement about (A), (B), (C) and (D) is:
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 50
Out of following isomeric forms of uracil, which one is present in RNA?
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 51
The reaction that occurs in a breath analyser, a device used to determine the alcohol level in a person's blood stream is
$$2K_2Cr_2O_7 + 8H_2SO_4 + 3C_2H_6O \rightarrow 2Cr_2(SO_4)_3 + 3C_2H_4O_2 + 2K_2SO_4 + 11H_2O$$
If the rate of appearance of $$Cr_2(SO_4)_3$$ is 2.67 mol min$$^{-1}$$ at a particular time, the rate of disappearance of $$C_2H_6O$$ at the same time is _________ mol min$$^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 52
1 kg of 0.75 molal aqueous solution of sucrose can be cooled up to $$-4°$$C before freezing. The amount of ice (in g) that will be separated out is _________. (Nearest integer) [Given : $$K_f(H_2O) = 1.86$$ K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 53
The number of $$f$$ electrons in the ground state electronic configuration of Np (Z = 93) is _________. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 54
The number of moles of NH$$_3$$, that must be added to 2 L of 0.80 M AgNO$$_3$$ in order to reduce the concentration of Ag$$^+$$ ions to $$5.0 \times 10^{-8}$$ M ($$K_{formation}$$ for $$[Ag(NH_3)_2]^+ = 1.0 \times 10^8$$) is _________. (Nearest integer)
[Assume no volume change on adding NH$$_3$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 55
When 10 mL of an aqueous solution of KMnO$$_4$$ was titrated in acidic medium, equal volume of 0.1M of an aqueous solution of ferrous sulphate was required for complete discharge of colour. The strength of KMnO$$_4$$ in grams per litre is _________ $$\times 10^{-2}$$. (Nearest integer) [Atomic mass of K = 39, Mn = 55, O = 16]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 56
The number of moles of CuO, that will be utilized in Dumas method for estimating nitrogen in a sample of 57.5 g of N, N-dimethylaminopentane is _________ $$\times 10^{-2}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 57
200 mL of 0.2 M HCl is mixed with 300 mL of 0.1 M NaOH. The molar heat of neutralization of this reaction is -57.1 kJ. The increase in temperature in °C of the system on mixing is $$x \times 10^{-2}$$. The value of x is _________. (Nearest integer)
[Given: Specific heat of water = 4.18 J g$$^{-1}$$ K$$^{-1}$$
Density of water = 1.00 g cm$$^{-3}$$]
(Assume no volume change on mixing)
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 58
The kinetic energy of an electron in the second Bohr orbit of a hydrogen atom is equal to $$\frac{h^2}{x \cdot m a_0^2}$$. The value of 10x is _________. ($$a_0$$ is radius of Bohr's orbit)
(Nearest integer)
[Given: $$\pi = 3.14$$]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 59
In Carius method for estimation of halogens, 0.2 g of an organic compound gave 0.188 g of AgBr. The percentage of bromine in the compound is _________. (Nearest integer)
[Atomic mass: Ag = 108, Br = 80]
789
456
123
0.-
Clear All
NTA JEE Main 27th August 2021 Shift 1 - Chemistry - Question 60
1 mol of an octahedral metal complex with formula MCl$$_3$$ . 2L on reaction with excess of AgNO$$_3$$ gives 1 mol of AgCl. The denticity of Ligand L is _________. (Integer answer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)



