NTA JEE Main 26th August 2021 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 31
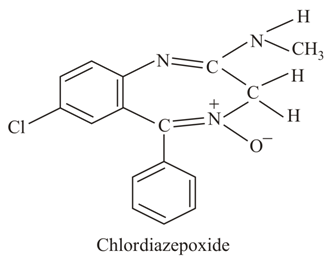
The class of drug to which chlordiazepoxide with above structure belongs is:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 32
The interaction energy of London forces between two particles is proportional to $$r^x$$, where r is the distance between the particles. The value of x is:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 33
The bond order and magnetic behaviour of $$O_2^-$$ ion are, respectively:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 34
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Heavy water is used for the study of reaction mechanism.
Reason (R): The rate of reaction for the cleavage of O-H bond is slower than that of O-D bond.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 35
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A): Barium carbonate is insoluble in water and is highly stable.
Reason (R): The thermal stability of the carbonates increases with increasing cationic size.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 36
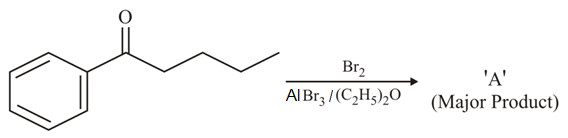
Consider the given reaction, the Product A is:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 37
Which one of the following compounds is not aromatic?
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 38
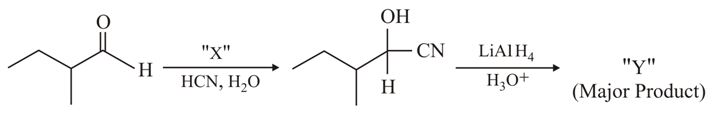
Consider the given reaction, Identify X and Y:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 39
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R)
Assertion (A) : Photochemical smog causes cracking of rubber.
Reason (R): Presence of ozone, nitric oxide, acrolein, formaldehyde and peroxyacetyl nitrate in photochemical smog makes it oxidizing.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 40
The sol given below with negatively charged colloidal particles is:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 41
Given below are two statements:
Statement I : Sphalerite is a sulphide ore of zinc and copper glance is a sulphide ore of copper.
Statement II : It is possible to separate two sulphide ores by adjusting proportion of oil to water or by using depressants in a froth flotation method.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 42
The number of non-ionisable hydrogen atoms present in the final product obtained from the hydrolysis of PCl$$_5$$ is:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 43
Chalcogen group elements are:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 44
Arrange the following Cobalt complexes in the order of increasing Crystal Field Stabilization Energy (CFSE) value.
Complexes:
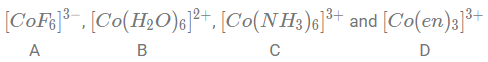
Choose the correct option:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 45
Indicate the complex/complex ion which did not show any geometrical isomerism:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 46
Which one of the following phenols does not give colour when condensed with phthalic anhydride in presence of conc. H$$_2$$SO$$_4$$?
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 47
The number of stereoisomers possible for 1,2-dimethyl cyclopropane is:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 48
Match List - I with List - II:
List - I (Chemical Reaction) List - II (Reagent used)
a. CH$$_3$$COOCH$$_2$$CH$$_3$$ $$\rightarrow$$ CH$$_3$$CH$$_2$$OH i. CH$$_3$$MgBr/H$$_3$$O$$^+$$ (one equivalent)
b. CH$$_3$$COOCH$$_3$$ $$\rightarrow$$ CH$$_3$$CHO ii. H$$_2$$SO$$_4$$/H$$_2$$O
c. CH$$_3$$C$$\equiv$$N $$\rightarrow$$ CH$$_3$$CHO iii. DIBAL-H/H$$_2$$O
d. iv. SnCl$$_2$$, HCl/H$$_2$$O
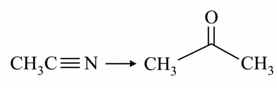
Choose the most appropriate match:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 49
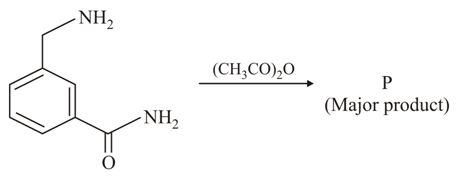
The Major Product in the above reaction is:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 50
Given below are two statements: one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Sucrose is a disaccharide and a non-reducing sugar.
Reason (R): Sucrose involves glycosidic linkage between $$C_1$$ of $$\beta$$-glucose and $$C_2$$ of $$\alpha$$-fructose.
Choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 51
100 mL of Na$$_3$$PO$$_4$$ solution contains 3.45 g of sodium. The molarity of the solution is _________ $$\times 10^{-2}$$ mol L$$^{-1}$$. (Nearest integer)
[Atomic Masses - Na : 23.0u, O : 16.0u, P : 31.0u]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 52
For water $$\Delta_{vap}H = 41$$ kJ mol$$^{-1}$$ at 373 K and 1 bar pressure. Assuming that water vapour is an ideal gas that occupies a much larger volume than liquid water, the internal energy change during evaporation of water is _________ (kJ mol$$^{-1}$$):
[Use: R = 8.3 J mol$$^{-1}$$ K$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 53
The equilibrium constant $$K_c$$ at 298 K for the reaction A + B $$\rightleftharpoons$$ C + D is 100. Starting with an equimolar solution with concentrations of A, B, C and D all equal to 1M, the equilibrium concentration of D is _________ $$\times 10^{-2}$$ M. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 54
In the sulphur estimation, 0.471 g of an organic compound gave 1.44 g of barium sulfate. The percentage of sulphur in the compound is _________ (Nearest integer) (Atomic Mass of Ba = 137u)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 55
The reaction rate for the reaction
$$[PtCl_4]^{2-} + H_2O \rightleftharpoons [Pt(H_2O)Cl_3]^- + Cl^-$$
was measured as a function of concentrations of different species. It was observed that
$$\frac{-d[PtCl_4]^{2-}}{dt} = 4.8 \times 10^{-5}[PtCl_4]^{2-} - 2.4 \times 10^{-3}[Pt(H_2O)Cl_3]^-][Cl^-]$$
where square brackets are used to denote molar concentrations.
The equilibrium constant K$$_c$$ = X (Nearest integer). Value of $$\frac{1}{X}$$ is _________
$$K_c = X$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 56
A chloro compound "A".
(i) forms aldehydes on ozonolysis followed by the hydrolysis.
(ii) when vaporized completely 1.53 g of A, gives 448 mL of vapour at STP. The number of carbon atoms in a molecule of compound A is _________
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 57
83 g of ethylene glycol dissolved in 625 g of water. The freezing point of the solution is _________ K. (Nearest integer)
[Use: Molal Freezing point depression constant of water = 1.86 K kg mol$$^{-1}$$
Freezing point of water = 273 K
Atomic masses: C : 12.0u, O : 16.0u, H : 1.0u]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 58
For the galvanic cell,
Zn(s) + Cu$$^{2+}$$(0.02M) $$\rightarrow$$ Zn$$^{2+}$$(0.04M) + Cu(s)
E$$_{cell}$$ = _________ $$\times 10^{-2}$$ V (Nearest integer)
[Use: E$$^0$$ Cu/Cu$$^{2+}$$ = $$-0.34$$ V, E$$_{Zn/Zn^{2+}}$$ = +0.76 V, $$\frac{2.303RT}{F}$$ = 0.059 V]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 59
The overall stability constant of the complex ion $$[Cu(NH_3)_4]^{2+}$$ is $$2.1 \times 10^{13}$$. The overall dissociation constant is $$y \times 10^{-14}$$. Then $$y$$ is _________ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 2 - Chemistry - Question 60
A metal surface is exposed to 500 nm radiation. The threshold frequency of the metal for photoelectric current is $$4.3 \times 10^{14}$$ Hz. The velocity of ejected electron is _________ $$\times 10^5$$ ms$$^{-1}$$ (Nearest integer)
[Use: h = $$6.63 \times 10^{-34}$$ Js, m$$_e$$ = $$9.0 \times 10^{-31}$$ kg]
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)



