NTA JEE Main 26th August 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 26th August 2021 Shift 1 - Question 31
Given below are two statements:
Statement I : According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increases with decrease in positive charges on the nucleus as there is no strong hold on the electron by the nucleus.
Statement II : According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increases with decrease in principle quantum number.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Question 32
Given below are two statements:
Statement I : In the titration between strong acid and weak base methyl orange is suitable as an indicator.
Statement II : For titration of acetic acid with NaOH phenolphthalein is not a suitable indicator.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Question 33
Which one of the following methods is most suitable for preparing deionized water?
NTA JEE Main 26th August 2021 Shift 1 - Question 34
What are the products formed in sequence when excess of CO$$_2$$ is passed in slaked lime?
NTA JEE Main 26th August 2021 Shift 1 - Question 35
The major product formed in the following reaction is: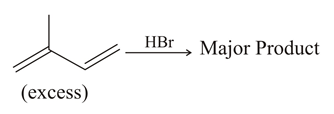
NTA JEE Main 26th August 2021 Shift 1 - Question 36
Excess of isobutane on reaction with Br$$_2$$ in presence of light at 125°C gives which one of the following, as the major product?
NTA JEE Main 26th August 2021 Shift 1 - Question 37
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with
(i) NaOH (ii) dil. HNO$$_3$$ (iii) AgNO$$_3$$
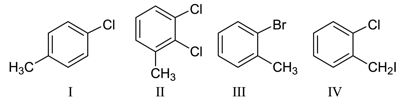
NTA JEE Main 26th August 2021 Shift 1 - Question 38
The conversion of hydroxyapatite occurs due to presence of F$$^-$$ ions in water. The correct formula of hydroxyapatite is:
NTA JEE Main 26th August 2021 Shift 1 - Question 39
Given below are two statements:
Statement I : Frenkel defects are vacancy as well as interstitial defects.
Statement II : Frenkel defect leads to colour in ionic solids due to presence of F-centres.
Choose the most appropriate answer for the statements from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Question 40
Given below are two statements:
Statement I : The limiting molar conductivity of KCl (strong electrolyte) is higher compared to that of CH$$_3$$COOH (weak electrolyte).
Statement II : Molar conductivity decreases with decrease in concentration of electrolyte.
In the light of the above statements, choose the most appropriate answer from the options given below:


.webp)
