NTA JEE Main 26th August 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 31
Given below are two statements:
Statement I : According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increases with decrease in positive charges on the nucleus as there is no strong hold on the electron by the nucleus.
Statement II : According to Bohr's model of an atom, qualitatively the magnitude of velocity of electron increases with decrease in principle quantum number.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 32
Given below are two statements:
Statement I : In the titration between strong acid and weak base methyl orange is suitable as an indicator.
Statement II : For titration of acetic acid with NaOH phenolphthalein is not a suitable indicator.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 33
Which one of the following methods is most suitable for preparing deionized water?
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 34
What are the products formed in sequence when excess of CO$$_2$$ is passed in slaked lime?
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 35
The major product formed in the following reaction is: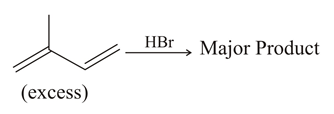
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 36
Excess of isobutane on reaction with Br$$_2$$ in presence of light at 125°C gives which one of the following, as the major product?
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 37
Among the following compounds I - IV, which one forms a yellow precipitate on reacting sequentially with
(i) NaOH (ii) dil. HNO$$_3$$ (iii) AgNO$$_3$$
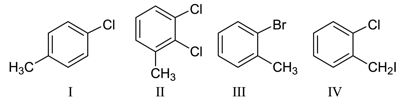
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 38
The conversion of hydroxyapatite occurs due to presence of F$$^-$$ ions in water. The correct formula of hydroxyapatite is:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 39
Given below are two statements:
Statement I : Frenkel defects are vacancy as well as interstitial defects.
Statement II : Frenkel defect leads to colour in ionic solids due to presence of F-centres.
Choose the most appropriate answer for the statements from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 40
Given below are two statements:
Statement I : The limiting molar conductivity of KCl (strong electrolyte) is higher compared to that of CH$$_3$$COOH (weak electrolyte).
Statement II : Molar conductivity decreases with decrease in concentration of electrolyte.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 41
Which one of the following is correct for the adsorption of a gas at a given temperature on a solid surface?
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 42
Given below are two statements.
Statement I: The choice of reducing agents for metals extraction can be made by using the Ellingham diagram, a plot of $$\Delta G$$ vs temperature.
Statement II: The value of $$\Delta S$$ increases from left to right in the Ellingham diagram.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 43
The incorrect statement is:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 44
Which one of the following when dissolved in water gives coloured solution in nitrogen atmosphere?
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 45
Which one of the following complexes is violet in colour?
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 46
The correct options for the products A and B of the following reactions are: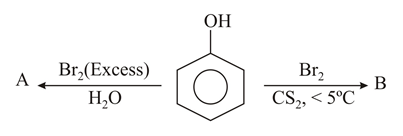
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 47
The major products formed in the following reaction sequence A and B are: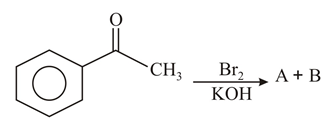
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 48
The major product formed in the following reaction is: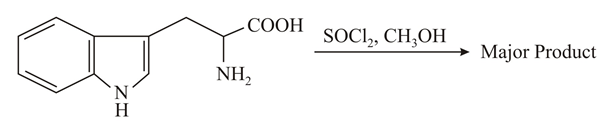
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 49
The correct sequential addition of reagents in the preparation of 3-nitrobenzoic acid from benzene is:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 50
The polymer formed on heating Novolac with formaldehyde is:
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 51
An aqueous KCl solution of density 1.20 g mL$$^{-1}$$ has a molality of 3.30 mol kg$$^{-1}$$. The molarity of the solution in mol L$$^{-1}$$ is _________ (The Nearest integer)
[Molar mass of KCl = 74.5 g]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 52
AB$$_3$$ is an interhalogen T-shaped molecule. The number of lone pairs of electrons on A is _________ (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 53
The Born-Haber cycle for KCl is evaluated with the following data:
$$\Delta_f H^{\ominus}$$ for KCl = $$-436.7$$ kJ mol$$^{-1}$$; $$\Delta_{sub} H^{\ominus}$$ for K = 89.2 kJ mol$$^{-1}$$;
$$\Delta_{ionization} H^{\ominus}$$ for K = 419.0 kJ mol$$^{-1}$$; $$\Delta_{electron gain} H^{\ominus}$$ for Cl$$_{(g)}$$ = $$-348.6$$ kJ mol$$^{-1}$$
$$\Delta_{bond} H^{\ominus}$$ for Cl$$_2$$ = 243.0 kJ mol$$^{-1}$$
The magnitude of lattice enthalpy of KCl in kJ mol$$^{-1}$$ is _________ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 54
The OH$$^-$$ concentration in a mixture of 5.0 mL of 0.0504 M NH$$_4$$Cl and 2 mL of 0.0210 M NH$$_3$$ solution is $$x \times 10^{-6}$$ M. The value of $$x$$ is _________ (Nearest integer) [Given $$K_w = 1 \times 10^{-14}$$ and $$K_b = 1.8 \times 10^{-5}$$]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 55
The ratio of number of water molecules in Mohr's salt and potash alum is _________ $$\times 10^{-1}$$. (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 56
Of the following four aqueous solutions, the total number of those solutions whose freezing point is lower than that of 0.10 M C$$_2$$H$$_5$$OH is _________ (Integer answer)
(i) 0.10 M Ba$$_3$$(PO$$_4$$)$$_2$$
(ii) 0.10 M Na$$_2$$SO$$_4$$
(iii) 0.10 M KCl
(iv) 0.10 M Li$$_3$$PO$$_4$$
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 57
These are physical properties of an element:
(A) Sublimation enthalpy
(B) Ionisation enthalpy
(C) Hydration enthalpy
(D) Electron gain enthalpy
The total number of above properties that affect the reduction potential is _________ (Integer answer)
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 58
The following data was obtained for chemical reaction given below at 975 K.
$$2NO_{(g)} + 2H_{2(g)} \rightarrow N_{2(g)} + 2H_2O_{(g)}$$
[NO] = 8 $$\times 10^{-5}$$, [H$$_2$$] = 8 $$\times 10^{-5}$$, Rate = 7 $$\times 10^{-9}$$
[NO] = 24 $$\times 10^{-5}$$, [H$$_2$$] = 8 $$\times 10^{-5}$$, Rate = 2.1 $$\times 10^{-8}$$
[NO] = 24 $$\times 10^{-5}$$, [H$$_2$$] = 32 $$\times 10^{-5}$$, Rate = 8.4 $$\times 10^{-8}$$
The order of the reaction with respect to NO is _________ [Integer answer]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 59
The number of 4f electrons in the ground state electronic configuration of Gd$$^{2+}$$ is _________ [Atomic number of Gd = 64]
789
456
123
0.-
Clear All
NTA JEE Main 26th August 2021 Shift 1 - Chemistry - Question 60
The total number of negative charge in the tetrapeptide, Gly-Glu-Asp-Tyr, at pH 12.5 will be _________ (Integer answer)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)



