NTA JEE Main 25th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 25th July 2022 Shift 2 - Question 51
$$56.0 \text{ L}$$ of nitrogen gas is mixed with excess of hydrogen gas and it is found that $$20 \text{ L}$$ of ammonia gas is produced. The volume of unused nitrogen gas is found to be ______ L.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 52
When the excited electron of a H atom from $$n = 5$$ drops to the ground state, the maximum number of emission lines observed are ______.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 53
The sum of number of lone pairs of electrons present on the central atoms of $$XeO_3$$, $$XeOF_4$$ and $$XeF_6$$ is ______.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 54
A sealed flask with a capacity of $$2 \text{ dm}^3$$ contains $$11 \text{ g}$$ of propane gas. The flask is so weak that it will burst if the pressure becomes $$2 \text{ MPa}$$. The minimum temperature at which the flask will burst is ______ °C. [Nearest integer]
(Given: $$R = 8.3 \text{ J K}^{-1} \text{ mol}^{-1}$$. Atomic masses of C and H are $$12u$$ and $$1u$$ respectively.) (Assume that propane behaves as an ideal gas.)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 55
While performing a thermodynamics experiment, a student made the following observations:
$$HCl + NaOH \rightarrow NaCl + H_2O \quad \Delta H = -57.3 \text{ kJ mol}^{-1}$$
$$CH_3COOH + NaOH \rightarrow CH_3COONa + H_2O \quad \Delta H = -55.3 \text{ kJ mol}^{-1}$$
The enthalpy of ionization of $$CH_3COOH$$ as calculated by the student is ______ $$\text{kJ mol}^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 56
The separation of two coloured substances was done by paper chromatography. The distances travelled by solvent front, substance A and substance B from the base line are $$3.25 \text{ cm}$$, $$2.08 \text{ cm}$$ and $$1.05 \text{ cm}$$ respectively. The ratio of $$R_f$$ values of A to B is ______ (Answer the nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 57
The total number of monobromo derivatives formed by the alkanes with molecular formula $$C_5H_{12}$$ is ______ (excluding stereo isomers).
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 58
For the decomposition of azomethane $$CH_3N_2CH_3(g) \rightarrow CH_3CH_3(g) + N_2$$, a first order reaction, the variation in partial pressure with time at $$600 \text{ K}$$ is given as shown.
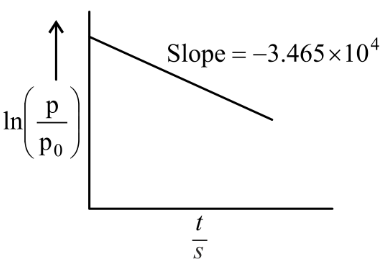
The half life of the reaction is ______ $$\times 10^{-5}$$ s.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 59
The spin-only magnetic moment value of $$M^{3+}$$ ion (in gaseous state) from the pairs $$Cr^{3+}/Cr^{2+}$$, $$Mn^{3+}/Mn^{2+}$$, $$Fe^{3+}/Fe^{2+}$$ and $$Co^{3+}/Co^{2+}$$ that has negative standard electrode potential, is ______ B.M.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2022 Shift 2 - Question 60
A sample of $$4.5 \text{ mg}$$ of an unknown monohydric alcohol, $$R-OH$$ was added to methylmagnesium iodide. A gas is evolved and is collected and its volume measured to be $$3.1 \text{ mL}$$. The molecular weight of the unknown alcohol is ______ g/mol.
789
456
123
0.-
Clear All


