NTA JEE Main 25th July 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 25th July 2022 Shift 2 - Question 31
The first ionization enthalpies of $$Be$$, $$B$$, $$N$$ and $$O$$ follow the order
NTA JEE Main 25th July 2022 Shift 2 - Question 32
Match List I with List II

Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 2 - Question 33
$$K_{a1}$$, $$K_{a2}$$ and $$K_{a3}$$ are the respective ionization constants for the following reactions (a), (b) and (c).
(a) $$H_2C_2O_4 \rightleftharpoons H^+ + HC_2O_4^-$$
(b) $$HC_2O_4^- \rightleftharpoons H^+ + C_2O_4^{2-}$$
(c) $$H_2C_2O_4 \rightleftharpoons 2H^+ + C_2O_4^{2-}$$
The relationship between $$K_{a1}$$, $$K_{a2}$$ and $$K_{a3}$$ is given as
NTA JEE Main 25th July 2022 Shift 2 - Question 34
In base vs. Acid titration, at the end point methyl orange is present as
NTA JEE Main 25th July 2022 Shift 2 - Question 35
High purity ($$> 99.95\%$$) dihydrogen is obtained by
NTA JEE Main 25th July 2022 Shift 2 - Question 36
The correct order of density is
NTA JEE Main 25th July 2022 Shift 2 - Question 37
Arrange the following in decreasing acidic strength.
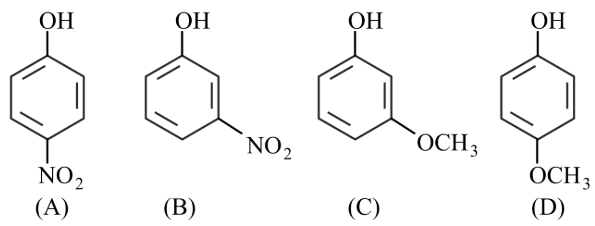
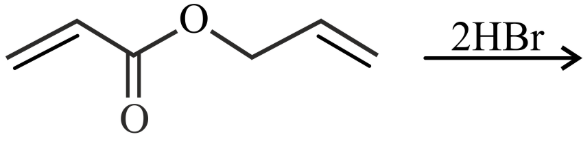
NTA JEE Main 25th July 2022 Shift 2 - Question 38
Major product of the following reaction is
NTA JEE Main 25th July 2022 Shift 2 - Question 39
Match List I with List II.

Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 2 - Question 40
Two solutions A and B are prepared by dissolving $$1 \text{ g}$$ of non-volatile solutes X and Y respectively in $$1 \text{ kg}$$ of water. The ratio of depression in freezing points for A and B is found to be $$1:4$$. The ratio of molar masses of X and Y is


