NTA JEE Main 25th July 2022 Shift 1
For the following questions answer them individually
NTA JEE Main 25th July 2022 Shift 1 - Question 31
$$SO_2Cl_2$$ on reaction with excess of water results into acidic mixture
$$SO_2Cl_2 + 2H_2O \rightarrow H_2SO_4 + 2HCl$$
16 moles of $$NaOH$$ is required for the complete neutralisation of the resultant acidic mixture. The number of moles of $$SO_2Cl_2$$ used is
NTA JEE Main 25th July 2022 Shift 1 - Question 32
Which of the following sets of quantum numbers is not allowed?
NTA JEE Main 25th July 2022 Shift 1 - Question 33
The IUPAC nomenclature of an element with electronic configuration $$[Rn] 5f^{14} 6d^1 7s^2$$ is
NTA JEE Main 25th July 2022 Shift 1 - Question 34
$$20 \text{ mL}$$ of $$0.1 \text{ M } NH_4OH$$ is mixed with $$40 \text{ mL}$$ of $$0.05 \text{ M HCl}$$. The pH of the mixture is nearest to:
(Given: $$K_b(NH_4OH) = 1 \times 10^{-5}$$, $$\log 2 = 0.30$$, $$\log 3 = 0.48$$, $$\log 5 = 0.69$$, $$\log 7 = 0.84$$, $$\log 11 = 1.04$$)
NTA JEE Main 25th July 2022 Shift 1 - Question 35
The reaction of $$H_2O_2$$ with potassium permanganate in acidic medium leads to the formation of mainly
NTA JEE Main 25th July 2022 Shift 1 - Question 36
Choose the correct order of density of the alkali metals
NTA JEE Main 25th July 2022 Shift 1 - Question 37
Match List - I with List - II
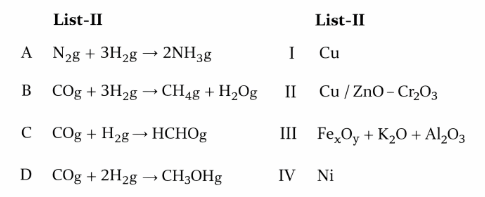
Choose the correct answer from the options given below
NTA JEE Main 25th July 2022 Shift 1 - Question 38
The geometry around boron in the product 'B' formed from the following reaction is
$$BF_3 + NaH \xrightarrow{450K} A + NaF$$
$$A + NMe_3 \rightarrow B$$
NTA JEE Main 25th July 2022 Shift 1 - Question 39
A compound 'A' on reaction with 'X' and 'Y' produces the same major product but different by-products 'a' and 'b'. Oxidation of 'a' gives a substance produced by ants.
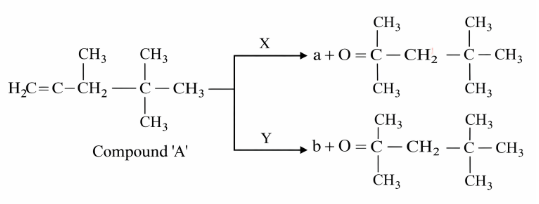
'X' and 'Y' respectively are
NTA JEE Main 25th July 2022 Shift 1 - Question 40
The photochemical smog does not generally contain


