NTA JEE Main 25th July 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 25th July 2021 Shift 2 - Question 51
The number of significant figures in 0.00340 is _________
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 52
An accelerated electron has a speed of $$5 \times 10^6$$ ms$$^{-1}$$ with an uncertainty of 0.02%. The uncertainty in finding its location while in motion is $$x \times 10^{-9}$$ m. The value of $$x$$ is _________. (Nearest integer)
[Use mass of electron = $$9.1 \times 10^{-31}$$ kg, h = $$6.63 \times 10^{-34}$$ Js, $$\pi$$ = 3.14]
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 53
An LPG cylinder contains gas at a pressure of 300 kPa at 27°C. The cylinder can withstand the pressure of $$1.2 \times 10^6$$ Pa. The room in which the cylinder is kept catches fire. The minimum temperature at which the bursting of cylinder will take place is _________ °C. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 54
A system does 200 J of work and at the same time absorbs 150 J of heat. The magnitude of the change in internal energy is _________J. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 55
Assuming that Ba(OH)$$_2$$ is completely ionised in aqueous solution under the given conditions the concentration of H$$_3$$O$$^+$$ ions in 0.005M aqueous solution of Ba(OH)$$_2$$ at 298 K is _________ $$\times 10^{-12}$$ mol L$$^{-1}$$. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 56
0.8 g of an organic compound was analysed by Kjeldahl's method for the estimation of nitrogen. If the percentage of nitrogen in the compound was found to be 42%, then _________ mL of 1M H$$_2$$SO$$_4$$ would have been neutralized by the ammonia evolved during the analysis.
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 57
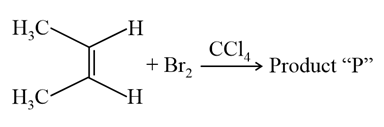
Consider the above chemical reaction. The total number of stereoisomers possible for Product "P" is _________
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 58
When 3.00 g of a substance X' is dissolved in 100 g of CCl$$_4$$, it raises the boiling point by 0.60 K. The molar mass of the substance 'X' is _________ g mol$$^{-1}$$. (Nearest integer).
[Given K$$_b$$ for CCl$$_4$$ is 5.0 K kg mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 59
For a chemical reaction A $$\rightarrow$$ B, it was found that concentration of B is increased by 0.2 mol L$$^{-1}$$ in 30 min. The average rate of the reaction is _________ $$\times 10^{-1}$$ mol L$$^{-1}$$ h$$^{-1}$$. (in nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 25th July 2021 Shift 2 - Question 60
Number of electrons present in 4f orbital of Ho$$^{3+}$$ ion is _________ (Given Atomic No. of Ho = 67)
789
456
123
0.-
Clear All


