NTA JEE Main 25th July 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 25th July 2021 Shift 2 - Question 31
The spin only magnetic moments (in BM) for free Ti$$^{3+}$$, V$$^{2+}$$ and Sc$$^{3+}$$ ions respectively are (At.No. Sc : 21, Ti : 22, V : 23)
NTA JEE Main 25th July 2021 Shift 2 - Question 32
The ionic radii of F$$^-$$ and O$$^{2-}$$ respectively are 1.33 A and 1.4A, while the covalent radius of N is 0.74A. The correct statement for the ionic radius of N$$^{3-}$$ from the following is:
NTA JEE Main 25th July 2021 Shift 2 - Question 33
In the following the correct bond order sequence is:
NTA JEE Main 25th July 2021 Shift 2 - Question 34
Identify the species having one $$\pi$$-bond and maximum number of canonical forms from the following:
NTA JEE Main 25th July 2021 Shift 2 - Question 35
Identify the process in which change in the oxidation state is five:
NTA JEE Main 25th July 2021 Shift 2 - Question 36
Which one of the following metals forms interstitial hydride easily?
NTA JEE Main 25th July 2021 Shift 2 - Question 37
Match List I with List II:
List-I (Elements) List-II (Properties)
(a) Li (i) Poor water solubility of I$$^-$$ salt
(b) Na (ii) Most abundant element in cell fluid
(c) K (iii) Bicarbonate salt used in fire extinguisher
(d) Cs (iv) Carbonate salt decomposes easily on heating
Choose the correct answer from the options given below:
NTA JEE Main 25th July 2021 Shift 2 - Question 38
The correct decreasing order of densities of the following compounds is: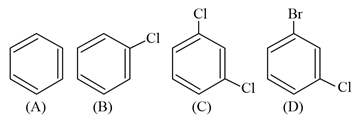
NTA JEE Main 25th July 2021 Shift 2 - Question 39
Which among the following is the strongest acid?
NTA JEE Main 25th July 2021 Shift 2 - Question 40
Given below are two statements:
Statement I : Chlorofluoro carbons breakdown by radiation in the visible energy region and release chlorine gas in the atmosphere which then reacts with stratospheric ozone.
Statement II : Atmospheric ozone reacts with nitric oxide to give nitrogen and oxygen gases, which add to the atmosphere.
For the above statements choose the correct answer from the options given below:


