NTA JEE Main 25th January 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 25th January 2023 Shift 2 - Question 51
Number of hydrogen atoms per molecule of a hydrocarbon A having 85.8% carbon is (Given: Molar mass of A = 84 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 52
The number of given orbitals which have electron density along the axis is
p$$_x$$, p$$_y$$, p$$_z$$, d$$_{xy}$$, d$$_{yz}$$, d$$_{xz}$$, d$$_z^2$$, d$$_{x^2-y^2}$$
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 53
28.0 L of CO$$_2$$ is produced on complete combustion of 16.8 L gaseous mixture of ethene and methane at 25°C and 1 atm. Heat evolved during the combustion process is _____ kJ
Given: $$\Delta H_C$$(CH$$_4$$) = -900 kJ mol$$^{-1}$$
$$\Delta H_C$$(C$$_2$$H$$_4$$) = -1400 kJ mol$$^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 54
The number of pairs of the solution having the same value of the osmotic pressure from the following is
(Assume 100% ionization)
A. 0.500 M C$$_2$$H$$_5$$OH(aq) and 0.25 M KBr(aq)
B. 0.100 M K$$_4$$[Fe(CN)$$_6$$](aq) and 0.100 M FeSO$$_4$$(NH$$_4$$)$$_2$$SO$$_4$$(aq)
C. 0.05 M K$$_4$$[Fe(CN)$$_6$$](aq) and 0.25 M NaCl(aq)
D. 0.15 M NaCl(aq) and 0.1 M BaCl$$_2$$(aq)
E. 0.02 M KCl.MgCl$$_2$$.6H$$_2$$O(aq) and 0.05 M KCl(aq)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 55
Pt(s)|H$$_2$$(g)(1 bar)|H$$^+$$(aq)(1M)||M$$^{3+}$$(aq), M$$^+$$(aq)|Pt(s)
The E$$_{cell}$$ for the given cell is 0.1115 V at 298 K
When $$\frac{[M^+(aq)]}{[M^{3+}(aq)]} = 10^a$$
The value of a is _____
Given: E$$^0_{M^{3+}/M^+}$$ = 0.2 V
$$\frac{2.303RT}{F}$$ = 0.059 V
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 56
A first order reaction has the rate constant, k = 4.6 $$\times 10^{-3}$$ s$$^{-1}$$. The number of correct statement/s from the following is/are Given: log 3 = 0.48.
A. Reaction completes in 1000 s.
B. The reaction has a half-life of 500 s.
C. The time required for 10% completion is 25 times the time required for 90% completion.
D. The degree of dissociation is equal to $$(1 - e^{-kt})$$.
E. The rate and the rate constant have the same unit.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 57
Based on the given figure, the number of correct statement/s is/are
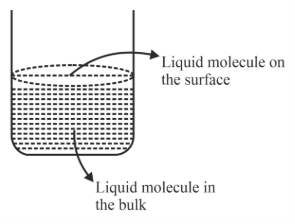
A. Surface tension is the outcome of equal attractive and repulsion forces acting on the liquid molecule in bulk.
B. Surface tension is due to uneven forces acting on the molecules present on the surface.
C. The molecule in the bulk can never come to the liquid surface.
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 58
The number of incorrect statement/s from the following is/are
A. Water vapours are adsorbed by anhydrous calcium chloride.
B. There is a decrease in surface energy during adsorption.
C. As the adsorption proceeds, $$\Delta$$H becomes more and more negative.
D. Adsorption is accompanied by decrease in entropy of the system.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 59
Total number of moles of AgCl precipitated on addition of excess of AgNO$$_3$$ to one mole each of the following complexes [Co(NH$$_3$$)$$_4$$Cl$$_2$$]Cl, [Ni(H$$_2$$O)$$_6$$]Cl$$_2$$, [Pt(NH$$_3$$)$$_2$$Cl$$_2$$] and [Pd(NH$$_3$$)$$_4$$]Cl$$_2$$ is
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Question 60
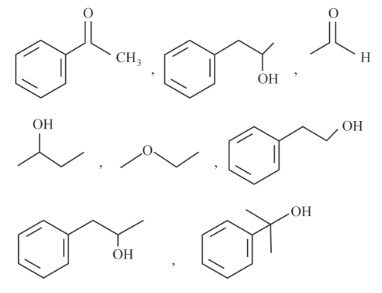
Number of compounds giving (i) red colouration with ceric ammonium nitrate and also (ii) positive iodoform test from the following is _____
789
456
123
0.-
Clear All


