NTA JEE Main 25th January 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 31
Statement I: Dipole moment is a vector quantity and by convention it is depicted by a small arrow with tail on the negative centre and head pointing towards the positive centre.
Statement II: The crossed arrow of the dipole moment symbolizes the direction of the shift of charges in the molecules.
In the light of the above statements, choose the most appropriate answer:
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 32
When the hydrogen ion concentration [H$$^+$$] changes by a factor of 1000, the value of pH of the solution
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 33
Match List I with List II
A. Cobalt catalyst I. (H$$_2$$ + Cl$$_2$$) production
B. Syngas II. Water gas production
C. Nickel catalyst III. Coal gasification
D. Brine solution IV. Methanol production
Choose the correct answer from the options given below:-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 34
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: The alkali metals and their salts impart characteristic colour to reducing flame.
Reason R: Alkali metals can be detected using flame tests.
In the light of the above statements, choose the most appropriate answer from the options given below
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 35
Which one among the following metals is the weakest reducing agent?
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 36
Which of the following represents the correct order of metallic character of the given elements?
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 37
Given below are two statements,one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Carbon forms two important oxides CO and CO$$_2$$. CO is neutral whereas CO$$_2$$ is acidic in nature.
Reason R: CO$$_2$$ can combine with water in a limited way to form carbonic acid, while CO is sparingly soluble in water.
In the light of the above statements, choose the most appropriate answer from the options given below :-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 38
Match List I with List II.
List I List II.
A. Propanamine and N-Methylethanamine I. Metamers
B. Hexan-2-one and Hexan-3-one II. Positional isomers
C. Ethanamide and Hydroxyethanimine III. Functional isomers
D. o-nitrophenol and pnitrophenol IV. Tautomers
Choose the correct answer from the options given below :-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 39
The isomeric deuterated bromide with molecular formula C$$_4$$H$$_8$$DBr having two chiral carbon atoms is
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 40
What is the mass ratio of ethylene glycol (C$$_2$$H$$_6$$O$$_2$$, molar mass = 62 g/mol) required for making 500 g of 0.25 molal aqueous solution and 250 mL of 0.25 molar aqueous solution?
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 41
Given below are two statements:-
Statement I: In froth floatation method a rotating paddle agitates the mixture to drive air out of it.
Statement II: Iron pyrites are generally avoided for extraction of iron due to environmental reasons.
In the light of the above statements, choose the correct answer:
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 42
A chloride salt solution acidified with dil. HNO$$_3$$ gives a curdy white precipitate, [A], on addition of AgNO$$_3$$. [A] on treatment with NH$$_4$$OH gives a clear solution, B.
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 43
Potassium dichromate acts as a strong oxidizing agent in acidic solution. During this process, the oxidation state changes from
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 44
Match List I with List II
List I List II
Coordination entity Wavelength of light absorbed in nm
A. [CoCl(NH$$_3$$)$$_5$$]$$^{2+}$$ I. 310
B. [Co(NH$$_3$$)$$_6$$]$$^{3+}$$ II. 475
C. [Co(CN)$$_6$$]$$^{3-}$$ III. 535
D. [Cu(H$$_2$$O)$$_4$$]$$^{2+}$$ IV. 600
Choose the correct answer from the options given below :-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 45
Find out the major product from the following reaction.
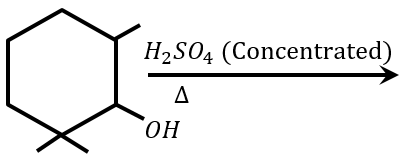
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 46
'A' in the given reaction is
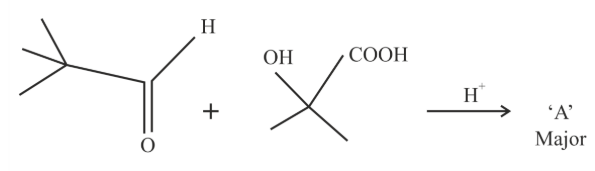
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 47
Given below are two statements,one is labelled as Assertion A and the other is labelled as Reason R
Assertion A: Butylated hydroxyl anisole when added to butter increases its shelf life.
Reason R: Butylated hydroxyl anisole is more reactive towards oxygen than food.
In the light of the above statements, choose the most appropriate answer from the options given below:-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 48
Match List I with List II
List I (Amines) List II (pKb)
A. Aniline I. 3.25
B. Ethanamine II. 3.00
C. N-Ethylethanamine III. 9.38
D. N,N-Diethylethanamine IV. 3.29
Choose the correct answer from the options given below:-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 49
Match List I with List II
List I (Name of polymer) List II (Uses)
A. Glyptal I. Flexible pipes
B. Neoprene II. Synthetic wool
C. Acrilan III. Paints and Lacquers
D. LDP IV. Gaskets
Choose the correct answer from the options given below :-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 50
A. Ammonium salts produce haze in atmosphere.
B. Ozone gets produced when atmospheric oxygen reacts with chlorine radicals.
C. Polychlorinated biphenyls act as cleansing solvents.
D. 'Blue baby' syndrome occurs due to the presence of excess of sulphate ions in water.
Choose the correct answer from the options below:-
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 51
Number of hydrogen atoms per molecule of a hydrocarbon A having 85.8% carbon is (Given: Molar mass of A = 84 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 52
The number of given orbitals which have electron density along the axis is
p$$_x$$, p$$_y$$, p$$_z$$, d$$_{xy}$$, d$$_{yz}$$, d$$_{xz}$$, d$$_z^2$$, d$$_{x^2-y^2}$$
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 53
28.0 L of CO$$_2$$ is produced on complete combustion of 16.8 L gaseous mixture of ethene and methane at 25°C and 1 atm. Heat evolved during the combustion process is _____ kJ
Given: $$\Delta H_C$$(CH$$_4$$) = -900 kJ mol$$^{-1}$$
$$\Delta H_C$$(C$$_2$$H$$_4$$) = -1400 kJ mol$$^{-1}$$.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 54
The number of pairs of the solution having the same value of the osmotic pressure from the following is
(Assume 100% ionization)
A. 0.500 M C$$_2$$H$$_5$$OH(aq) and 0.25 M KBr(aq)
B. 0.100 M K$$_4$$[Fe(CN)$$_6$$](aq) and 0.100 M FeSO$$_4$$(NH$$_4$$)$$_2$$SO$$_4$$(aq)
C. 0.05 M K$$_4$$[Fe(CN)$$_6$$](aq) and 0.25 M NaCl(aq)
D. 0.15 M NaCl(aq) and 0.1 M BaCl$$_2$$(aq)
E. 0.02 M KCl.MgCl$$_2$$.6H$$_2$$O(aq) and 0.05 M KCl(aq)
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 55
Pt(s)|H$$_2$$(g)(1 bar)|H$$^+$$(aq)(1M)||M$$^{3+}$$(aq), M$$^+$$(aq)|Pt(s)
The E$$_{cell}$$ for the given cell is 0.1115 V at 298 K
When $$\frac{[M^+(aq)]}{[M^{3+}(aq)]} = 10^a$$
The value of a is _____
Given: E$$^0_{M^{3+}/M^+}$$ = 0.2 V
$$\frac{2.303RT}{F}$$ = 0.059 V
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 56
A first order reaction has the rate constant, k = 4.6 $$\times 10^{-3}$$ s$$^{-1}$$. The number of correct statement/s from the following is/are Given: log 3 = 0.48.
A. Reaction completes in 1000 s.
B. The reaction has a half-life of 500 s.
C. The time required for 10% completion is 25 times the time required for 90% completion.
D. The degree of dissociation is equal to $$(1 - e^{-kt})$$.
E. The rate and the rate constant have the same unit.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 57
Based on the given figure, the number of correct statement/s is/are
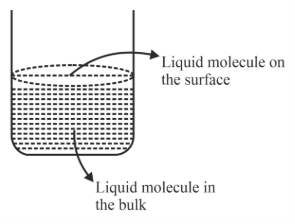
A. Surface tension is the outcome of equal attractive and repulsion forces acting on the liquid molecule in bulk.
B. Surface tension is due to uneven forces acting on the molecules present on the surface.
C. The molecule in the bulk can never come to the liquid surface.
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 58
The number of incorrect statement/s from the following is/are
A. Water vapours are adsorbed by anhydrous calcium chloride.
B. There is a decrease in surface energy during adsorption.
C. As the adsorption proceeds, $$\Delta$$H becomes more and more negative.
D. Adsorption is accompanied by decrease in entropy of the system.
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 59
Total number of moles of AgCl precipitated on addition of excess of AgNO$$_3$$ to one mole each of the following complexes [Co(NH$$_3$$)$$_4$$Cl$$_2$$]Cl, [Ni(H$$_2$$O)$$_6$$]Cl$$_2$$, [Pt(NH$$_3$$)$$_2$$Cl$$_2$$] and [Pd(NH$$_3$$)$$_4$$]Cl$$_2$$ is
789
456
123
0.-
Clear All
NTA JEE Main 25th January 2023 Shift 2 - Chemistry - Question 60
Number of compounds giving (i) red colouration with ceric ammonium nitrate and also (ii) positive iodoform test from the following is _____
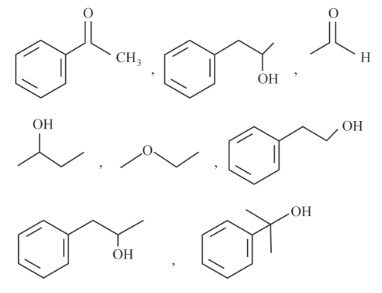
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




