NTA JEE Main 25th February 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 25th February 2021 Shift 1 - Question 31
Complete combustion of 1.80 g of an oxygen containing compound ($$C_xH_yO_z$$) gave 2.64 g of $$CO_2$$ and 1.08 g of $$H_2O$$. The percentage of oxygen in the organic compound is:
NTA JEE Main 25th February 2021 Shift 1 - Question 32
The plots of radial distribution functions for various orbitals of hydrogen atom against 'r' are given below. The correct plot for 3s orbital is:
NTA JEE Main 25th February 2021 Shift 1 - Question 33
According to molecular orbital theory, the species among the following that does not exist is:
NTA JEE Main 25th February 2021 Shift 1 - Question 34
The solubility of AgCN in a buffer solution of pH = 3 is x. The value of x is: [Assume: No cyano complex is formed; $$K_{sp}(AgCN) = 2.2 \times 10^{-16}$$ and $$K_a(HCN) = 6.2 \times 10^{-10}$$]
NTA JEE Main 25th February 2021 Shift 1 - Question 35
Which of the following equation depicts the oxidizing nature of $$H_2O_2$$?
NTA JEE Main 25th February 2021 Shift 1 - Question 36
The incorrect statement about $$B_2H_6$$ is:
NTA JEE Main 25th February 2021 Shift 1 - Question 37
Compound(s) which will liberate carbon dioxide with sodium bicarbonate solution is/are:
NTA JEE Main 25th February 2021 Shift 1 - Question 38
Identify A in the given chemical reaction.
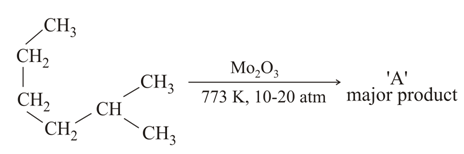
NTA JEE Main 25th February 2021 Shift 1 - Question 39
Which of the following reaction/s will not give $$p$$-aminoazobenzene?
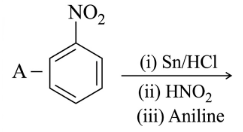
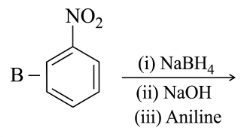
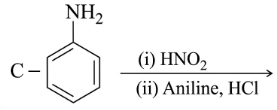
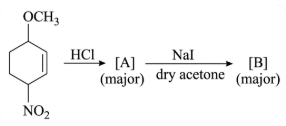
NTA JEE Main 25th February 2021 Shift 1 - Question 40
Identify A and B in the chemical reaction.