NTA JEE Main 25th April 2013 Online
For the following questions answer them individually
NTA JEE Main 25th April 2013 Online - Question 41
In which of the following exothermic reactions, the heat liberated per mole is the highest?
NTA JEE Main 25th April 2013 Online - Question 42
Given that: (i) $$\Delta_f H°$$ of $$N_2O$$ is 82 kJ mol$$^{-1}$$ (ii) Bond energies of $$N \equiv N$$, $$N = N$$, $$O = O$$ and $$N = O$$ are 946, 418, 498 and 607 kJ mol$$^{-1}$$ respectively. The resonance energy of $$N_2O$$ is :
NTA JEE Main 25th April 2013 Online - Question 43
The ratio $$\frac{K_p}{K_c}$$ for the reaction $$CO(g) + \frac{1}{2}O_2(g) \rightleftharpoons CO_2(g)$$ is:
NTA JEE Main 25th April 2013 Online - Question 44
What would be the pH of a solution obtained by mixing 5 g of acetic acid and 7.5 g of sodium acetate and making the volume equal to 500 mL? ($$K_a = 1.75 \times 10^{-5}$$, pK$$_a$$ = 4.76)
NTA JEE Main 25th April 2013 Online - Question 45
Which one of the following arrangements represents the correct order of the proton affinity of the given species :
NTA JEE Main 25th April 2013 Online - Question 46
Given
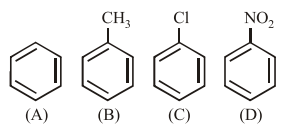
In the above compounds correct order of reactivity in electrophilic substitution reactions will be:
NTA JEE Main 25th April 2013 Online - Question 47
Copper crystallises in fcc with a unit length of 361 pm. What is the radius of copper atom?
NTA JEE Main 25th April 2013 Online - Question 48
The Gibbs energy for the decomposition of $$Al_2O_3$$ at 500°C is as follows :
$$\frac{2}{3}Al_2O_3 \rightarrow \frac{4}{3}Al + O_2$$, $$\Delta_r G = +940$$ kJ mol$$^{-1}$$
The potential difference needed for the electrolytic reduction of aluminium oxide at 500°C should be at least :
NTA JEE Main 25th April 2013 Online - Question 49
A solution of copper sulphate (CuSO$$_4$$) is electrolysed for 10 minutes with a current of 1.5 amperes. The mass of copper deposited at the cathode (at. mass of Cu = 63u) is :
NTA JEE Main 25th April 2013 Online - Question 50
A radioactive isotope having a half-life period of 3 days was received after 12 days. If 3 g of the isotope is left in the container, what would be the initial mass of the isotope?


