NTA JEE Main 24th June 2022 Shift 2
For the following questions answer them individually
NTA JEE Main 24th June 2022 Shift 2 - Question 51
At $$300$$ K, a sample of $$3.0$$ g of gas A occupies the same volume as $$0.2$$ g of hydrogen at $$200$$ K at the same pressure. The molar mass of gas A is ______ g mol$$^{-1}$$. (nearest integer) Assume that the behaviour of gases as ideal.
(Given: The molar mass of hydrogen ($$H_2$$) gas is $$2.0$$ g mol$$^{-1}$$.)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 52
$$PCl_5$$ dissociates as $$PCl_5(g) \rightleftharpoons PCl_3(g) + Cl_2(g)$$. $$5$$ moles of $$PCl_5$$ are placed in a $$200$$ litre vessel which contains $$2$$ moles of $$N_2$$ and is maintained at $$600$$ K. The equilibrium pressure is $$2.46$$ atm. The equilibrium constant $$K_p$$ for the dissociation of $$PCl_5$$ is ______ $$\times 10^{-3}$$. (nearest integer) (Given: $$R = 0.082$$ L atm K$$^{-1}$$ mol$$^{-1}$$; Assume ideal gas behaviour)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 53
Manganese (VI) has ability to disproportionate in acidic solution. The difference in oxidation states of two ions it forms in acidic solution is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 54
$$0.2$$ g of an organic compound was subjected to estimation of nitrogen by Dumas method in which volume of $$N_2$$ evolved (at STP) was found to be $$22.400$$ mL. The percentage of nitrogen in the compound is ______ [nearest integer] (Given: Molar mass of $$N_2$$ is $$28$$ g mol$$^{-1}$$, Molar volume of $$N_2$$ at STP: $$22.4$$ L)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 55
A company dissolves '$$x$$' amount of $$CO_2$$ at $$298$$ K in $$1$$ litre of water to prepare soda water. $$X = $$ ______ $$\times 10^{-3}$$ g. (nearest integer)
(Given: partial pressure of $$CO_2$$ at $$298$$ K $$= 0.835$$ bar. Henry's law constant for $$CO_2$$ at $$298$$ K $$= 1.67$$ kbar. Atomic mass of H, C and O is $$1, 12,$$ and $$6$$ g mol$$^{-1}$$, respectively)
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 56
The resistance of a conductivity cell containing $$0.01$$ MKCl solution at $$298$$ K is $$1750$$ $$\Omega$$. If the conductivity of $$0.01$$ MKCl solution at $$298$$ K is $$0.152 \times 10^{-3}$$ S cm$$^{-1}$$, then the cell constant of the conductivity cell is ______ $$\times 10^{-3}$$ cm$$^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 57
When $$200$$ mL of $$0.2$$ M acetic acid is shaken with $$0.6$$ g of wood charcoal, the final concentration of acetic acid after adsorption is $$0.1$$ M. The mass of acetic acid adsorbed per gram of carbon is ______ g.
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 58
(a) Baryte, (b) Galena, (c) Zinc blende and (d) Copper pyrites. How many of these minerals are sulphide based? ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 59
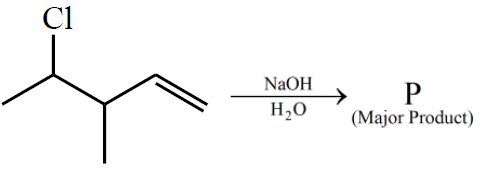
Consider the above reaction. The number of $$\pi$$ electrons present in the product 'P' is ______
789
456
123
0.-
Clear All
NTA JEE Main 24th June 2022 Shift 2 - Question 60
In alanylglycylleucylalanylvaline the number of peptide linkages is: ______
789
456
123
0.-
Clear All


