NTA JEE Main 24th January 2023 Shift 2
For the following questions answer them individually
NTA JEE Main 24th January 2023 Shift 2 - Question 31
What is the number of unpaired electron(s) in the highest occupied molecular orbital of the following species: N$$_2$$, N$$_2^+$$, O$$_2$$, O$$_2^+$$?
NTA JEE Main 24th January 2023 Shift 2 - Question 32
Choose the correct representation of conductometric titration of benzoic acid vs sodium hydroxide.
NTA JEE Main 24th January 2023 Shift 2 - Question 33
In which of the following reactions the hydrogen peroxide acts as a reducing agent?
NTA JEE Main 24th January 2023 Shift 2 - Question 34
Identify the correct statements about alkali metals.
A. The order of standard reduction potential (M$$^+$$ | M) for alkali metal ions is Na > Rb > Li.
B. CsI is highly soluble in water.
C. Lithium carbonate is highly stable to heat.
D. Potassium dissolved in concentrated liquid ammonia is blue and paramagnetic.
E. All alkali metal hydrides are ionic solids.
Choose the correct answer from the options given below
NTA JEE Main 24th January 2023 Shift 2 - Question 35
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Beryllium has less negative value of reduction potential compared to the other alkaline earth metals.
Reason R: Beryllium has large hydration energy due to small size of Be$$^{2+}$$ but relatively large value of atomisation enthalpy.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 24th January 2023 Shift 2 - Question 36
The number of s-electrons present in an ion with 55 protons in its unipositive state is
NTA JEE Main 24th January 2023 Shift 2 - Question 37
Which will undergo deprotonation most readily in basic medium?
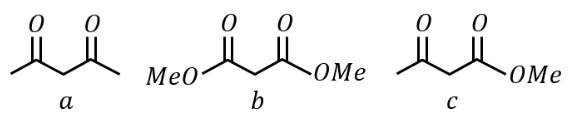
NTA JEE Main 24th January 2023 Shift 2 - Question 38
Given below are two statements:
Statement I:
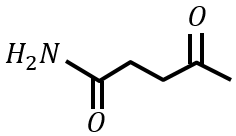
under Clemmensen reduction conditions will give
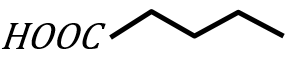
Statement II:
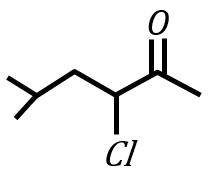
under Wolff-Kishner reduction condition will give
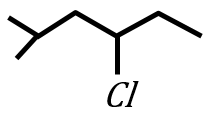
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 24th January 2023 Shift 2 - Question 39
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Benzene is more stable than hypothetical cyclohexatriene.
Reason R: The delocalized $$\pi$$ electron cloud is attracted more strongly by nuclei of carbon atoms.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 24th January 2023 Shift 2 - Question 40
The metal which is extracted by oxidation and subsequent reduction from its ore is:


