NTA JEE Main 24th January 2023 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 31
What is the number of unpaired electron(s) in the highest occupied molecular orbital of the following species: N$$_2$$, N$$_2^+$$, O$$_2$$, O$$_2^+$$?
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 32
Choose the correct representation of conductometric titration of benzoic acid vs sodium hydroxide.
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 33
In which of the following reactions the hydrogen peroxide acts as a reducing agent?
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 34
Identify the correct statements about alkali metals.
A. The order of standard reduction potential (M$$^+$$ | M) for alkali metal ions is Na > Rb > Li.
B. CsI is highly soluble in water.
C. Lithium carbonate is highly stable to heat.
D. Potassium dissolved in concentrated liquid ammonia is blue and paramagnetic.
E. All alkali metal hydrides are ionic solids.
Choose the correct answer from the options given below
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 35
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Beryllium has less negative value of reduction potential compared to the other alkaline earth metals.
Reason R: Beryllium has large hydration energy due to small size of Be$$^{2+}$$ but relatively large value of atomisation enthalpy.
In the light of the above statements, choose the most appropriate answer from the options given below.
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 36
The number of s-electrons present in an ion with 55 protons in its unipositive state is
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 37
Which will undergo deprotonation most readily in basic medium?
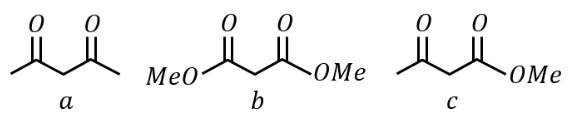
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 38
Given below are two statements:
Statement I:
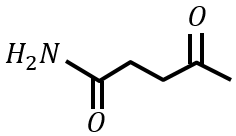
under Clemmensen reduction conditions will give
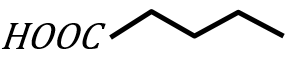
Statement II:
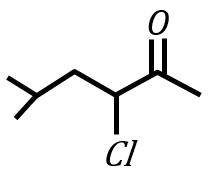
under Wolff-Kishner reduction condition will give
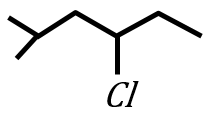
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 39
Given below are two statements, one is labelled as Assertion A and the other is labelled as Reason R.
Assertion A: Benzene is more stable than hypothetical cyclohexatriene.
Reason R: The delocalized $$\pi$$ electron cloud is attracted more strongly by nuclei of carbon atoms.
In the light of the above statements, choose the correct answer from the options given below:
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 40
The metal which is extracted by oxidation and subsequent reduction from its ore is:
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 41
Which one amongst the following are good oxidising agents?
(a) Sm$$^{2+}$$
(b) Ce$$^{2+}$$
(c) Ce$$^{4+}$$
(d) Tb$$^{4+}$$
Choose the most appropriate answer from the options below:
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 42
K$$_2$$Cr$$_2$$O$$_7$$ paper acidified with dilute H$$_2$$SO$$_4$$ turns green when exposed to
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 43
Which of the following cannot be explained by crystal field theory?
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 44
The hybridization and magnetic behaviour of cobalt ion in [Co(NH$$_3$$)$$_6$$]$$^{3+}$$ complex, respectively is
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 45
Find out the major products from the following reactions.
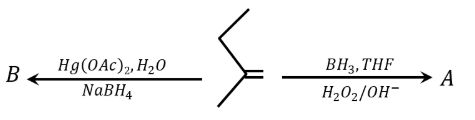
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 46
Given below are two statements:
Statement I: Pure Aniline and other arylamines are usually colourless.
Statement II: Arylamines get coloured on storage due to atmospheric reduction.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 47
Choose the correct colour of the product for the following reaction.
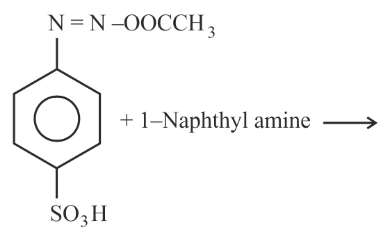
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 48
Correct statement is:
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 49
Match List I with List II
LIST I (Type) LIST II (Name)
A. Antifertility drug I. Norethindrone
B. Tranquilizer II. Meprobromate
C. Antihistamine III. Seldane
D. Antibiotic IV. Ampicillin
Choose the correct answer from the options given below:
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 50
Following figure shows spectrum of an ideal black body at four different temperatures. The number of correct statement/s from the following is _____.
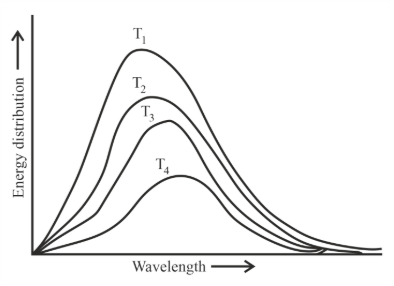
A. $$T_4 > T_3 > T_2 > T_1$$
B. The black body consists of particles performing simple harmonic motion.
C. The peak of the spectrum shifts to shorter wavelength as temperature increases.
D. $$\frac{T_1}{\nu_1} = \frac{T_2}{\nu_2} = \frac{T_3}{\nu_3}$$
E. The given spectrum could be explained using quantisation of energy
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 51
Sum of $$\pi$$-bonds present in peroxodisulphuric acid and pyrosulphuric acid is
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 52
The number of statement's, which are correct with respect to the compression of carbon dioxide from point (a) in the Andrews isotherm from the following is _____.
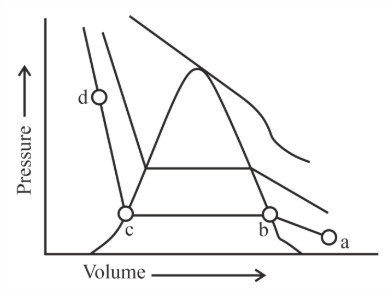
A. Carbon dioxide remains as a gas upto point (b)
B. Liquid carbon dioxide appears at point (c)
C. Liquid and gaseous carbon dioxide coexist between points (b) and (c)
D. As the volume decreases from (b) to (c), the amount of liquid decreases
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 53
One mole of an ideal monoatomic gas is subjected to changes as shown in the graph. The magnitude of the work done (by the system or on the system) is _____ J (nearest integer)
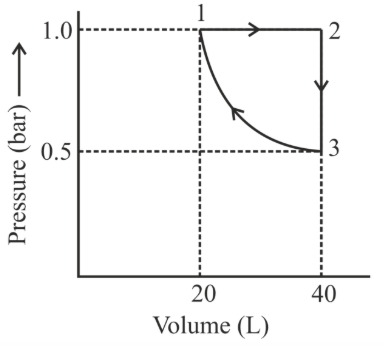
Given: log 2 = 0.3, ln 10 = 2.3
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 54
If the pKa of lactic acid is 5, then the pH of 0.005 M calcium lactate solution at 25°C is _____ $$\times 10^{-1}$$ (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 55
The Total pressure observed by mixing two liquid A and B is 350 mm Hg when their mole fractions are 0.7 and 0.3 respectively. The Total pressure becomes 410 mm Hg if the mole fractions are changed to 0.2 and 0.8 respectively for A and B. The vapour pressure of pure A is _____ mm Hg. (Nearest integer) Consider the liquids and solutions behave ideally
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 56
The number of units, which are used to express concentration of solutions from the following is _____.
(Mass percent, Mole, Mole fraction, Molarity, ppm, Molality.)
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 57
A student has studied the decomposition of a gas $$AB_3$$ at 25°C. He obtained the following data
| p (mm Hg) | 50 | 100 | 200 | 400 |
|---|---|---|---|---|
| Relative $$t_{1/2}$$ (s) | 4 | 2 | 1 | 0.5 |
The order of the reaction is
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 58
The number of statement/s which are the characteristics of physisorption is _____.
A. It is highly specific in nature
B. Enthalpy of adsorption is high
C. It decreases with increase in temperature
D. It results into unimolecular layer
E. No activation energy is needed
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 59
Maximum number of isomeric monochloro derivatives which can be obtained from 2,2,5,5-tetramethylhexane by chlorination is _____
789
456
123
0.-
Clear All
NTA JEE Main 24th January 2023 Shift 2 - Chemistry - Question 60
Total number of tripeptides possible by mixing of valine and proline is _____.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




