NTA JEE Main 24th February 2021 Shift 2
For the following questions answer them individually
NTA JEE Main 24th February 2021 Shift 2 - Question 51
The formula of a gaseous hydrocarbon which requires 6 times of its own volume of $$O_2$$ for complete oxidation and produces 4 times its own volume of $$CO_2$$ is $$C_xH_y$$. The value of y is ______.
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 52
1.86 g of aniline completely reacts to form acetanilide. 10% of the product is lost during purification. Amount of acetanilide obtained after purification (in g) is ______ $$\times 10^{-2}$$.
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 53
The volume occupied by 4.75 g of acetylene gas at 50°C and 740 mm Hg pressure is ______ L.
(Rounded off to the nearest integer)
[Given R = 0.0826 L atm K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 54
Assuming ideal behaviour, the magnitude of log K for the following reaction at 25°C is $$x \times 10^{-1}$$. The value of x is ______. (Integer answer)
$$3HC \equiv CH_{(g)} \rightleftharpoons C_6H_{6(l)}$$
[Given: $$\Delta_f G^{\circ}(HC \equiv CH) = -2.04 \times 10^5$$ J mol$$^{-1}$$; $$\Delta_f G^{\circ}(C_6H_6) = -1.24 \times 10^5$$ J mol$$^{-1}$$; R = 8.314 J K$$^{-1}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 55
The solubility product of $$PbI_2$$ is $$8.0 \times 10^{-9}$$. The solubility of lead iodide in 0.1 molar solution of lead nitrate is $$x \times 10^{-6}$$ mol/L. The value of x is ______ (Rounded off to the nearest integer)
[Given $$\sqrt{2} = 1.41$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 56
$$C_6H_6$$ freezes at 5.5°C. The temperature at which a solution of 10 g of $$C_4H_{10}$$ in 200 g of $$C_6H_6$$ freeze is ______ °C. (nearest integer value), (The molal freezing point depression constant of $$C_6H_6$$ is 5.12°C/m.)
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 57
The magnitude of the change in oxidising power of the $$MnO_4^- / Mn^{2+}$$ couple is $$x \times 10^{-4}$$ V if the $$H^+$$ concentration is decreased from 1 M to $$10^{-4}$$ M at 25°C. (Assume concentration of $$MnO_4^-$$ and $$Mn^{2+}$$ to be same on change in $$H^+$$ concentration). The value of x is ______. (Rounded off to the nearest integer)
[Given: $$\frac{2.303RT}{F} = 0.059$$]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 58
Sucrose hydrolyses in acid solution into glucose and fructose following first order rate law with a half-life of 3.33 h at 25°C. After 9 h, the fraction of sucrose remaining is f. The value of $$\log_{10}\left(\frac{1}{f}\right)$$ is ______ $$\times 10^{-2}$$.
(Rounded off to the nearest integer)
[Assume: ln10 = 2.303, ln 2 = 0.693]
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 59
Among the following allotropic forms of sulphur, the number of allotropic forms, which will show paramagnetism is ______.
(A) $$\alpha$$-sulphur
(B) $$\beta$$-sulphur
(C) $$S_2$$-form
789
456
123
0.-
Clear All
NTA JEE Main 24th February 2021 Shift 2 - Question 60
The total number of amines among the following which can be synthesized by Gabriel synthesis is ______.
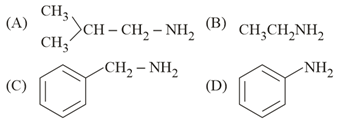
789
456
123
0.-
Clear All


