NTA JEE Main 24th February 2021 Shift 1
For the following questions answer them individually
NTA JEE Main 24th February 2021 Shift 1 - Question 31
Consider the elements Mg, Al, S, P and Si, the correct increasing order of their first ionisation enthalpy is:
NTA JEE Main 24th February 2021 Shift 1 - Question 32
Which of the following are isostructural pairs?
A. $$SO_4^{2-}$$ and $$CrO_4^{2-}$$
B. $$SiCl_4$$ and $$TiCl_4$$
C. $$NH_3$$ and $$NO_3^-$$
D. $$BCl_3$$ and $$BrCl_3$$
NTA JEE Main 24th February 2021 Shift 1 - Question 33
(A) $$HOCl + H_2O_2 \to H_3O^+ + Cl^- + O_2$$
(B) $$I_2 + H_2O_2 + 2OH^- \to 2I^- + 2H_2O + O_2$$
Choose the correct option.
NTA JEE Main 24th February 2021 Shift 1 - Question 34
$$Al_2O_3$$ was leached with alkali to get X. The solution of X on passing of gas Y, forms Z. X, Y and Z respectively are
NTA JEE Main 24th February 2021 Shift 1 - Question 35
Identify products A and B.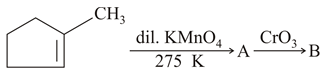
NTA JEE Main 24th February 2021 Shift 1 - Question 36
Which of the following compound gives pink colour on reaction with phthalic anhydride in conc. $$H_2SO_4$$ followed by treatment with NaOH?
NTA JEE Main 24th February 2021 Shift 1 - Question 37
In the following reaction, the reason why meta-nitro product also formed is:
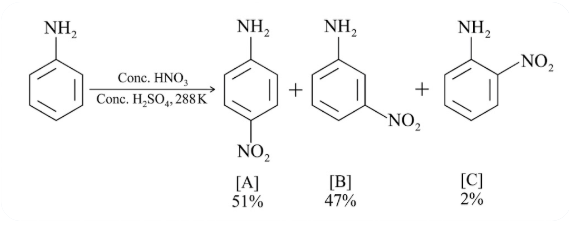
NTA JEE Main 24th February 2021 Shift 1 - Question 38
The gas released during anaerobic degradation of vegetation may lead to:
NTA JEE Main 24th February 2021 Shift 1 - Question 39
In Freundlich adsorption isotherm, slope of AB line is: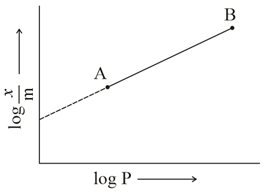
NTA JEE Main 24th February 2021 Shift 1 - Question 40
Which of the following ore is concentrated using group 1 cyanide salt?


