NTA JEE Main 22nd July 2021 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 31
Which one of the following statements for D.I. Mendeleeff, is incorrect?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 32
Match List-I with List-II
| (a) SF$$_4$$ | (i) sp$$^3$$d$$^2$$ |
| (b) IF$$_5$$ | (ii) d$$^2$$sp$$^3$$ |
| (c) NO$$_2^+$$ | (iii) sp$$^3$$d |
| (d) NH$$_4^+$$ | (iv) sp$$^3$$ |
| (v) sp |
Choose the correct answer from the options given below:
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 33
Match List-I with List-II
| (a) Ba | (i) Organic solvent soluble compounds |
| (b) Ca | (ii) Outer electronic configuration 6s$$^2$$ |
| (c) Li | (iii) Oxalate insoluble in water |
| (d) Na | (iv) Formation of very strong monoacidic base |
Choose the correct answer from the options given below:
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 34
Given below are the statements about diborane
(a) Diborane is prepared by the oxidation of NaBH$$_4$$ with I$$_2$$
(b) Each boron atom is in sp$$^2$$ hybridized state
(c) Diborane has one bridged 3 centre-2-electron bond
(d) Diborane is a planar molecule
The option with correct statement(s) is
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 35
Which purification technique is used for high boiling organic liquid compound (decomposes near its boiling point)?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 36
Which of the following compounds does not exhibit resonance?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 37
Which of the following molecules does not show stereo isomerism?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 38
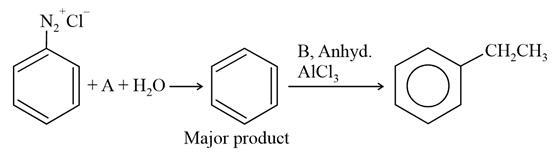
In the chemical reactions given above A and B respectively are:
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 39
The water having more dissolved O$$_2$$ is:
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 40
Which one of the following 0.06 M aqueous solutions has lowest freezing point?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 41
Isotope(s) of hydrogen which emits low energy $$\beta^-$$ particles with $$t_{1/2}$$ value > 12 years is/are
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 42
When silver nitrate solution is added to potassium iodide solution then the sol produced is:
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 43
Sulphide ion is soft base and its ores are common for metals
(a) Pb (b) Al (c) Ag (d) Mg
Choose the correct answer from the options given below
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 44
Which one of the following group-15 hydride is the strongest reducing agent?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 45
The set having ions which are coloured and paramagnetic both is -
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 46
Which of the following compounds will provide a tertiary alcohol on reaction with excess of CH$$_3$$MgBr followed by hydrolysis?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 47
An organic compound A (C$$_6$$H$$_6$$O) gives dark green colouration with ferric chloride. On treatment with CHCl$$_3$$ and KOH, followed by acidification gives compound B. Compound B can also be obtained from compound C on reaction with pyridinium chlorochromate (PCC). Identify A, B and C.
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 48
Which one of the following reactions does not occur?
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 49
Match List-I with List-II :
| (a) Chloroprene | (i)
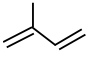 |
| (b) Neoprene | (ii)
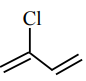 |
| (c) Acrylonitrile | (iii)
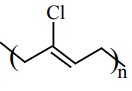 |
| (d) Isoprene | (iv) CH$$_2$$=CH-CN |
Choose the correct answer from the options given
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 50
Thiamine and pyridoxine are also known respectively as:
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 51
Methylation of 10 g of benzene gave 9.2 g of toluene. Calculate the percentage yield of toluene (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 52
Number of electrons that Vanadium (Z = 23) has in p-orbitals is equal to:
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 53
If the standard molar enthalpy change for combustion of graphite powder is $$-2.48 \times 10^2$$ kJ mol$$^{-1}$$, the amount of heat generated on combustion of 1 g of graphite powder in kJ (Nearest integer):
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 54
Value of K$$_P$$ for the equilibrium reaction N$$_2$$O$$_4$$(g) $$\rightleftharpoons$$ 2NO$$_2$$(g) at 288 K is 47.9. The K$$_C$$ for this reaction at same temperature is (Nearest integer)
(R = 0.083 L bar K$$^{-1}$$ mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 55
The number of acyclic structural isomers (including geometrical isomers) for pentene are ___.
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 56
A copper complex crystallising in a CCP lattice with a cell edge of 0.4518 nm has been revealed by employing X-ray diffraction studies. The density of a copper complex is found to be 7.62 g cm$$^{-3}$$. The molar mass of copper complex is ___ gmol$$^{-1}$$
(Nearest integer): [Given : N$$_A$$ = 6.022 $$\times 10^{23}$$ mol$$^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 57
If the concentration of glucose (C$$_6$$H$$_{12}$$O$$_6$$) in blood is 0.72 gL$$^{-1}$$, the molarity of glucose in blood is ___ $$\times 10^{-3}$$M (Nearest integer):
[Given : Atomic mass of C = 12, H = 1, O = 16 u]
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 58
Assume a cell with the following reaction Cu$$_{(s)}$$ + 2Ag$$^+$$(1 $$\times 10^{-3}$$M) $$\to$$ Cu$$^{2+}$$(0.250M) + 2Ag$$_{(s)}$$
E$$^\circ_{cell}$$ = 2.97 V
E$$_{cell}$$ for the above reaction is ___ V.
(Nearest integer)
[Given : log 2.5 = 0.3979, T = 298 K]
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 59
N$$_2$$O$$_{5(g)} \to$$ 2NO$$_{2(g)}$$ + $$\frac{1}{2}$$O$$_{2(g)}$$
In the above first order reaction the initial concentration of N$$_2$$O$$_5$$ is $$2.40 \times 10^{-2}$$ mol L$$^{-1}$$ at 318 K. The concentration of N$$_2$$O$$_5$$ after 1 hour was $$1.60 \times 10^{-2}$$ mol L$$^{-1}$$. The rate constant of the reaction at 318 K is ___ $$\times 10^{-3}$$ min$$^{-1}$$ (Nearest integer):
[Given : log 3 = 0.477, log 5 = 0.699]
789
456
123
0.-
Clear All
NTA JEE Main 22nd July 2021 Shift 1 - Chemistry - Question 60
The total number of unpaired electrons present in [Co(NH$$_3$$)$$_6$$]Cl$$_2$$ and [Co(NH$$_3$$)$$_6$$]Cl$$_3$$ is ___.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




