NTA JEE Main 2025 April 8th Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 51
In a first order decomposition reaction, the time taken for the decomposition of reactant to one fourth and one eighth of its initial concentration are $$t_1$$ and $$t_2$$ (s), respectively. The ratio $$t_1/t_2$$ will :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 52
Match the LIST-I with LIST-II.
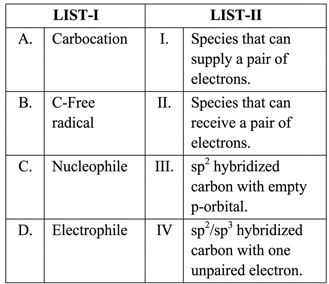
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 53
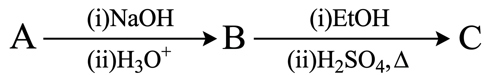
$$A \xrightarrow{(i) NaOH, (ii) H_3O^+} B \xrightarrow{(i) EtOH, (ii) H_2SO_4, \Delta} C$$
'A' shows positive Lassaign's test for N and its molar mass is 121. 'B' gives effervescence with aq. NaHCO$$_3$$. 'C' gives fruity smell. Identify A, B and C.
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 54
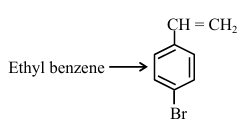
Choose the correct set of reagents for the following conversion:
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 55
1,2-dibromocyclooctane is treated with
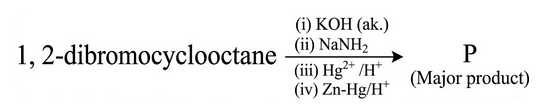
'P' is :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 56
Given below are two statements :
Statement I : A homoleptic octahedral complex, formed using monodentate ligands, will not show stereoisomerism.
Statement II : cis- and trans-platin are heteroleptic complexes of Pd.
Choose the correct answer from the options given below.
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 57
The atomic number of the element from the following with lowest 1st ionisation enthalpy is :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 58
Which of the following binary mixture does not show the behaviour of minimum boiling azeotropes?
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 59
$$HA(aq) \rightleftharpoons H^+(aq) + A^-(aq)$$. The freezing point depression of a 0.1 m aqueous solution of a monobasic weak acid HA is 0.20°C. The dissociation constant for the acid is : (Given: $$K_f(H_2O) = 1.8$$ K kg mol$$^{-1}$$, molality $$\equiv$$ molarity)
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 60
What is the correct IUPAC name of
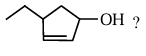
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 61
The correct decreasing order of spin only magnetic moment values (BM) of $$Cu^+$$, $$Cu^{2+}$$, $$Cr^{2+}$$ and $$Cr^{3+}$$ ions is :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 62
Which one of the following reactions will not lead to the desired ether formation in major proportion? (iso-Bu $$\Rightarrow$$ isobutyl, sec-Bu $$\Rightarrow$$ sec-butyl, nPr $$\Rightarrow$$ n-propyl, $$^t$$Bu $$\Rightarrow$$ tert-butyl, Et $$\Rightarrow$$ ethyl)
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 63
On combustion 0.210 g of an organic compound containing C, H and O gave 0.127 g $$H_2O$$ and 0.307 g $$CO_2$$. The percentages of hydrogen and oxygen in the given organic compound respectively are :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 64
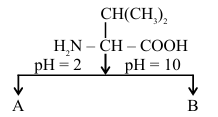
Choose the correct option for structures of A and B, respectively.
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 65
Correct statements for an element with atomic number 9 are :
A. There can be 5 electrons for which $$m_s = +\frac{1}{2}$$ and 4 electrons for which $$m_s = -\frac{1}{2}$$
B. There is only one electron in $$p_z$$ orbital
C. The last electron goes to orbital with $$n = 2$$ and $$l = 1$$
D. The sum of angular nodes of all the atomic orbitals is 1.
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 66
The number of species from the following that are involved in $$sp^3d^2$$ hybridization is : $$[Co(NH_3)_6]^{3+}$$, $$SF_6$$, $$[CrF_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Mn(CN)_6]^{3-}$$, and $$[MnCl_6]^{3-}$$
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 67
Match the LIST-I with LIST-II

Choose the correct answer from the options given below :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 68
When
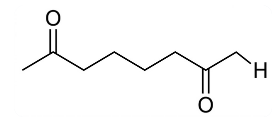
undergoes intramolecular aldol condensation, the major product formed is :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 69
Match the LIST-I with LIST-II
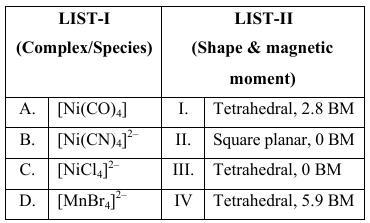
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 70
Given below are two statements :
Statement I : $$H_2Se$$ is more acidic than $$H_2Te$$.
Statement II : $$H_2Se$$ has higher bond enthalpy for dissociation than $$H_2Te$$.
In the light of the above statements, choose the correct answer from the options given below.
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 71
Resonance in $$X_2Y$$ can be represented as
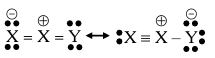
The enthalpy of formation of X_2Y\left( X\equiv X(g)+\frac{1}{2}Y=Y(g)\rightarrow X_2 Y(g)\right) is 80 kJ mol$$^{-1}$$. The magnitude of resonance energy of $$X_2Y$$ is _____ kJ mol$$^{-1}$$ (nearest integer value). Given: Bond energies of $$X \equiv X$$, $$X = X$$, $$Y = Y$$ and $$X = Y$$ are 940, 410, 500 and 602 kJ mol$$^{-1}$$ respectively.
valence X : 3, Y : 2
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 72
The energy of an electron in first Bohr orbit of H-atom is -13.6 eV. The magnitude of energy value of electron in the first excited state of $$Be^{3+}$$ is _____ eV. (nearest integer value)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 73
20 mL of sodium iodide solution gave 4.74 g silver iodide when treated with excess of silver nitrate solution. The molarity of the sodium iodide solution is _____ M. (Given : Na = 23, I = 127, Ag = 108, N = 14, O = 16 g mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 74
The equilibrium constant for decomposition of $$H_2O(g)$$
$$H_2O(g) \rightleftharpoons H_2(g) + \frac{1}{2}O_2(g)$$ ($$\Delta G^\circ = 92.34$$ kJ mol$$^{-1}$$) is $$8.0 \times 10^{-3}$$ at 2300 K and total pressure at equilibrium is 1 bar. Under this condition, the degree of dissociation ($$\alpha$$) of water is _____ $$\times 10^{-2}$$. (nearest integer value)
[Assume $$\alpha$$ is negligible with respect to 1]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 8th Shift 2 - Chemistry - Question 75
Consider the following half cell reaction: $$Cr_2O_7^{2-}(aq) + 6e^- + 14H^+(aq) \to 2Cr^{3+}(aq) + 7H_2O(l)$$. The reaction was conducted with the ratio of $$\frac{[Cr^{3+}]^2}{[Cr_2O_7^{2-}]} = 10^{-6}$$. The pH value at which the EMF of the half cell will become zero is _____ . (nearest integer value) [Given : standard half cell reduction potential $$E^0_{Cr_{2}O^{2-}_{7},H^{+}/Cr^{3+}} = 1.33$$ V, $$\frac{2.303RT}{F} = 0.059$$ V]
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




