NTA JEE Main 2025 April 7th Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 51
Given below are two statements :
Statement (I) : On hydrolysis, oligo peptides give rise to fewer number of $$\alpha$$-amino acids while proteins give rise to a large number of $$\beta$$-amino acids.
Statement (II) : Natural proteins are denatured by acids which convert the water soluble form of fibrous proteins to their water insoluble form.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 52
Mixture of 1 g each of chlorobenzene, aniline and benzoic acid is dissolved in 50 mL ethyl acetate and placed in a separating funnel, 5 M NaOH (30 mL) was added in the same funnel. The funnel was shaken vigorously and then kept aside. The ethyl acetate layer in the funnel contains :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 53
The hydration energies of $$K^+$$ and $$Cl^-$$ are $$-x$$ and $$-y$$ kJ/mol respectively. If lattice energy of KCl is $$-z$$ kJ/mol, then the heat of solution of KCl is :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 54
$$A(g) \to B(g) + C(g)$$ is a first order reaction.
The reaction was started with reactant A only. Which of the following expression is correct for rate constant k?
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 55
"P" is an optically active compound with molecular formula $$C_6H_{12}O$$. When "P" is treated with 2,4-dinitrophenylhydrazine, it gives a positive test. However, in presence of Tollens reagent, "P" gives a negative test. Predict the structure of "P".
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 56
Choose the incorrect trend in the atomic radii (r) of the elements :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 57
Match List-I with List-II .
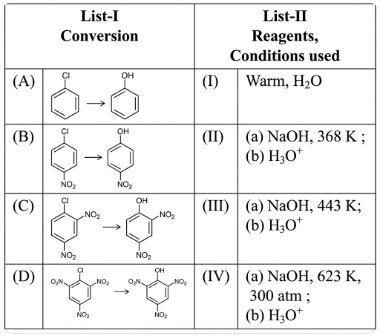
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 58
The correct statement amongst the following is :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 59
Liquid A and B form an ideal solution. The vapour pressure of pure liquids A and B are 350 and 750 mm Hg respectively at the same temperature. If $$x_A$$ and $$x_B$$ are the mole fraction of A and B in solution while $$y_A$$ and $$y_B$$ are the mole fraction of A and B in vapour phase then :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 60
'X' is the number of acidic oxides among $$VO_2$$, $$V_2O_3$$, $$CrO_3$$, $$V_2O_5$$ and $$Mn_2O_7$$. The primary valency of cobalt in $$[Co(H_2NCH_2CH_2NH_3)_3]_2(SO_4)_3$$ is Y. The value of X + Y is :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 61
The descending order of basicity of following amines is :
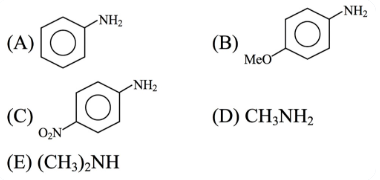
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 62
Match List-I with List-II.
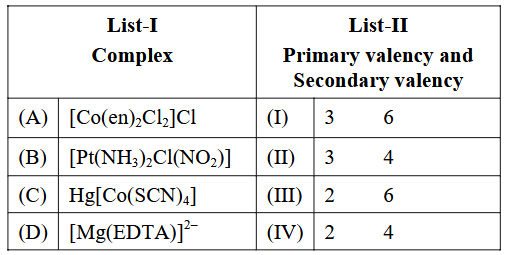
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 63
Match List-I with List-II.
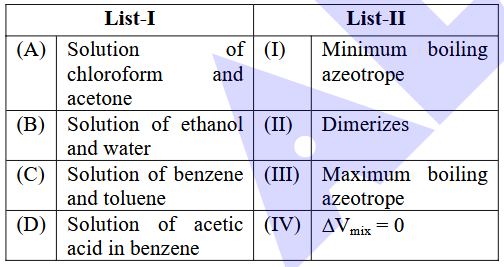
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 64
In $$SO_2$$, $$NO_2^-$$ and $$N_3^-$$ the hybridizations at the central atom are respectively :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 65
The number of unpaired electrons responsible for the paramagnetic nature of the following species are respectively : $$[Fe(CN)_6]^{3-}$$, $$[FeF_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Mn(CN)_6]^{3-}$$
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 66
The number of optically active products obtained from the complete ozonolysis of the given compound is :
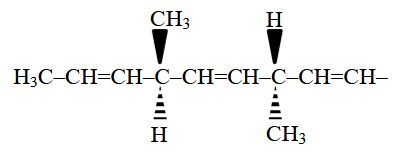
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 67
Given below are two statements :
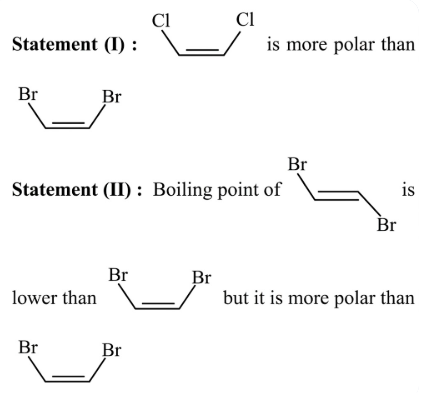
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 68
The extra stability of half-filled subshell is due to :
(A) Symmetrical distribution of electrons
(B) Smaller coulombic repulsion energy
(C) The presence of electrons with the same spin in non-degenerate orbitals
(D) Larger exchange energy
(E) Relatively smaller shielding of electrons by one another
Identify the correct statements
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 69
The correct statements from the following are :
(A) $$Tl^{3+}$$ is a powerful oxidising agent
(B) $$Al^{3+}$$ does not get reduced easily
(C) Both $$Al^{3+}$$ and $$Tl^{3+}$$ are very stable in solution
(D) $$Tl^+$$ is more stable than $$Tl^{3+}$$
(E) $$Al^{3+}$$ and $$Tl^+$$ are highly stable
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 70
Given below are two statements :
1 M aqueous solution of each of $$Cu(NO_3)_2$$, $$AgNO_3$$, $$Hg_2(NO_3)_2$$; $$Mg(NO_3)_2$$ are electrolysed using inert electrodes.
Given : $$E_{Ag^{+}/Ag}^{\theta} = 0.80V, E_{Hg_{2}^{2+}/Hg}^{\theta} = 0.79V,$$
$$E_{Cu^{2+}/Cu}^{\theta} = 0.24V$$ and $$E_{Mg^{2+}/Mg}^{\theta} = -2.37V$$
Statement (I) : With increasing voltage, the sequence of deposition of metals on the cathode will be Ag, Hg and Cu
Statement (II) : Magnesium will not be deposited at cathode instead oxygen gas will be evolved at the cathode.
In the light of the above statement, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 71
Only litre buffer solution was prepared by adding 0.10 mol each of $$NH_3$$ and $$NH_4Cl$$ in deionised water. The change in pH on addition of 0.05 mol of HCl to the above solution is _____ $$\times 10^{-2}$$.(Nearest integer)
(Given : $$pK_b$$ of $$NH_3$$ = 4.745 and $$\log_{10}3 = 0.477$$)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 72
In Dumas' method 292 mg of an organic compound released 50 mL of nitrogen gas ($$N_2$$) at 300 K temperature and 715 mm Hg pressure. The percentage composition of 'N' in the organic compound is _____ %.(Nearest integer)
(Aqueous tension at 300 K = 15 mm Hg)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 73
Butane reacts with oxygen to produce carbon dioxide and water following the equation given below
$$C_4H_{10}(g) + \frac{13}{2}O_2(g) \to 4CO_2(g) + 5H_2O(l)$$.
If 174.0 kg of butane is mixed with 320.0 kg of $$O_2$$, the volume of water formed in litres is _____.(Nearest integer)
[Given : (a) Molar mass of C, H, O are 12, 1, 16 g $$mol^{-1}$$ respectively, (b) Density of water = 1 g $$mL^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 74
The number of paramagnetic metal complex species among $$[Co(NH_3)_6]^{3+}$$, $$[Co(C_2O_4)_3]^{3-}$$, $$[MnCl_6]^{3-}$$, $$[Mn(CN)_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Fe(CN)_6]^{3-}$$ and $$[FeF_6]^{3-}$$ with same number of unpaired electrons is _____.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 7th Shift 2 - Chemistry - Question 75
Identify the structure of the final product (D) in the following sequence of reactions:
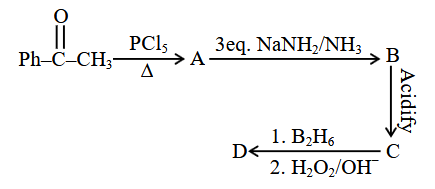
Total number of $$sp^2$$ hybridised carbon atoms in product D is _____.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




