NTA JEE Main 2025 April 4th Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 51
The correct order of basicity for the following molecules is :
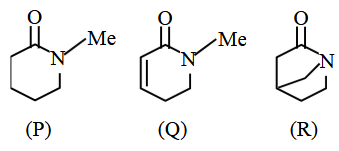
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 52
The incorrect relationship in the following pairs in relation to ionisation enthalpies is :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 53
Which among the following compounds give yellow solid when reacted with NaOI/NaOH?
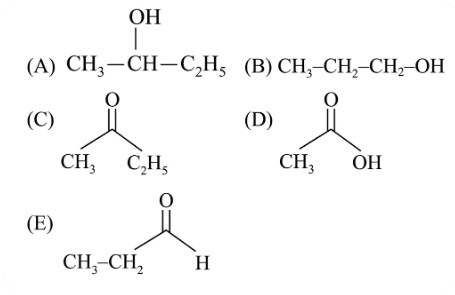
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 54
A dipeptide, "x" on complete hydrolysis gives "y" and "z". "y" on treatment with aq. HNO$$_2$$ produces lactic acid. On the other hand "z" on heating gives a cyclic molecule.
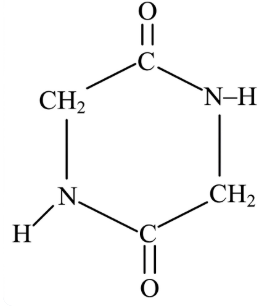
Based on the information given, the dipeptide X is:
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 55
In which pairs, the first ion is more stable than the second?
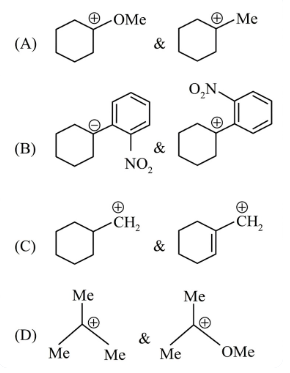
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 56
Given below are two statements :
Statement (I) : Alcohols are formed when alkyl chlorides are treated with aqueous potassium hydroxide by elimination reaction.
Statement (II) : In alcoholic potassium hydroxide, alkyl chlorides form alkenes by abstracting the hydrogen from the $$\beta$$-carbon.
In the light of the above statements, choose the most appropriate answer from the option given below :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 57
Given below are two statements :
Statement (I) : Molal depression constant $$K_f$$ is given by $$\frac{M_1 R T_f}{\Delta S_{fus}}$$, where symbols have their usual meaning.
Statement (II) : $$K_f$$ for benzene is less than the $$K_f$$ for water.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 58
The IUPAC name of the following compound is :
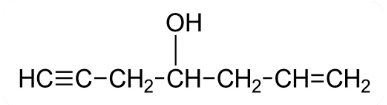
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 59
Match List-I (Separation of) with List-II (Separation Technique) :
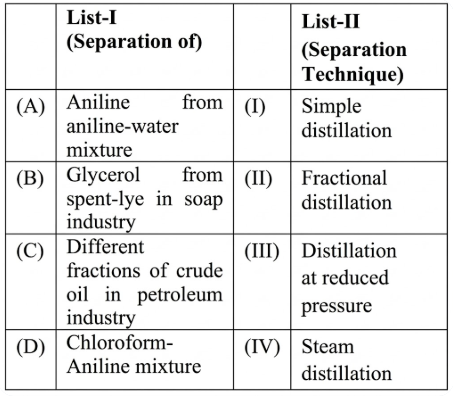
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 60
A toxic compound "A" when reacted with NaCN in aqueous acidic medium yields an edible cooking component and food preservative 'B'. "B" is converted to "C" by diborane and can be used as an additive to petrol to reduce emission. "C" upon reaction with oleum at $$140^{\circ}$$ C yields an inhalable anesthetic "D". Identify "A", "B", "C" and "D", respectively.
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 61
The correct order of $$[FeF_6]^{3-}$$, $$[CoF_6]^{3-}$$, $$[Ni(CO)_4]$$ and $$[Ni(CN)_4]^{2-}$$ complex species based on the number of unpaired electrons present is :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 62
Consider the given data :
(a) $$HCl(g) + 10H_2O(l) \rightarrow HCl \cdot 10H_2O$$, $$\Delta H = -69.01$$ kJ mol$$^{-1}$$
(b) $$HCl(g) + 40H_2O(l) \rightarrow HCl \cdot 40H_2O$$, $$\Delta H = -72.79$$ kJ mol$$^{-1}$$
Choose the correct statement :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 63
Consider the ground state of chromium atom (Z = 24). How many electrons are with Azimuthal quantum number $$l = 1$$ and $$l = 2$$ respectively?
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 64
Given below are two statements :
Statement (I) : The first ionisation enthalpy of group 14 elements is higher than the corresponding elements of group 13.
Statement (II) : Melting points and boiling points of group 13 elements are in general much higher than those the corresponding elements of group 14.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 65
Consider the following plots of log of rate constant k (log k) vs $$\frac{1}{T}$$ for three different reactions. The correct order of activation energies of these reactions is :
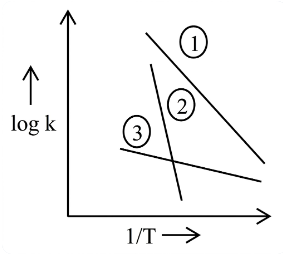
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 66
'X' is the number of electrons in $$t_{2g}$$ orbitals of the most stable complex ion among $$[Fe(NH_3)_6]^{3+}$$, $$[Fe(Cl)_6]^{3-}$$, $$[Fe(C_2O_4)_3]^{3-}$$ and $$[Fe(H_2O)_6]^{3+}$$. The nature of oxide of vanadium of the type $$V_2O_x$$ is:
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 67
The elements of Group 13 with highest and lowest first ionisation enthalpies are respectively:
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 68
Consider the following molecule (X). The structure of X is
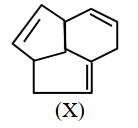
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 69
Given below are two statements:
Statement (I) : for

, all three possible structures may be drawn.
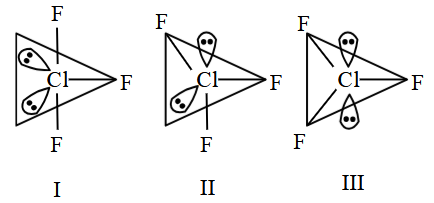
Statement (II) : Structure III is most stable, as the orbitals having the lone pairs are axial, where the $$lp-bp$$ repulsion is minimum.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 70
Half life of zero order reaction $$A \rightarrow$$ product is 1 hour, when initial concentration of reaction is 2.0 mol L$$^{-1}$$. The time required to decrease concentration of A from 0.50 to 0.25 mol L$$^{-1}$$ is:
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 71
Sea water, which can be considered as a 6 molar (6 M) solution of NaCl, has a density of 2 g mL$$^{-1}$$. The concentration of dissolved oxygen (O$$_2$$) in sea water is 5.8 ppm. Then the concentration of dissolved oxygen (O$$_2$$) in sea water, is $$x \times 10^{-4}$$ m. (Nearest integer)
Given: Molar mass of $$NaCl$$ is 58.5 g $$mol^{-1}$$
Molar mass of $$O_{2}$$ is 32 g $$mol^{-1}$$
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 72
The amount of calcium oxide produced on heating 150 kg limestone (75% pure) is ______ kg. (Nearest integer)
Given : Molar mass (in g $$mol^{-1}$$) of $$Ca-40, O-16, C-12$$
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 73
A metal complex with a formula $$MCl_4 \cdot 3NH_3$$ is involved in $$sp^3d^2$$ hybridisation. It upon reaction with excess of $$AgNO_3$$ solution gives 'x' moles of AgCl. Consider 'x' is equal to the number of lone pairs of electron present in central atom of $$BrF_5$$. Then the number of geometrical isomers exhibited by the complex is ______.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 74
The molar conductance of an infinitely dilute solution of ammonium chloride was found to be 185 S cm$$^2$$ mol$$^{-1}$$ and the ionic conductance of hydroxyl and chloride ions are 170 and 70 S cm$$^2$$ mol$$^{-1}$$, respectively. If molar conductance of 0.02 M solution of ammonium hydroxide is 85.5 S cm$$^2$$ mol$$^{-1}$$, its degree of dissociation is given by $$x \times 10^{-1}$$. The value of x is ______. (Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 4th Shift 2 - Chemistry - Question 75
x mg of $$Mg(OH)_2$$ (molar mass = 58) is required to be dissolved in 1.0 L of water to produce a pH of 10.0 at 298 K. The value of x is ____ mg. (Nearest integer)
(Given : $$Mg(OH)_{2}$$ is assumed to dissociate completely in $$H_{2}O$$)
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




