NTA JEE Main 2025 April 3rd Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 51
40 mL of a mixture of $$CH_3COOH$$ and HCl (aqueous solution) is titrated against 0.1 M NaOH conductometrically. Which of the following statement is correct?
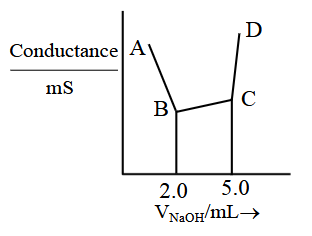
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 52
10 mL of 2 M NaOH solution is added to 20 mL of 1 M HCl solution kept in a beaker. Now, 10 mL of this mixture is poured into a volumetric flask of 100 mL containing 2 moles of HCl and made the volume upto the mark with distilled water. The solution in this flask is:
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 53
Fat soluble vitamins are:
A. Vitamin $$B_1$$
B. Vitamin C
C. Vitamin E
D. Vitamin $$B_{12}$$
E. Vitamin K
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 54
Match the LIST-I with LIST-II.
| LIST-I (Family) | LIST-II (Symbol) | ||
|---|---|---|---|
| A. | Pnicogen (group 15) | I. | Ts |
| B. | Chalcogen | II. | Og |
| C. | Halogen | III. | Lv |
| D. | Noble gas | IV. | Mc |
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 55
For electron in '2s' and '2p' orbitals, the orbital angular momentum values, respectively are:
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 56
Compounds that should not be used as primary standards in titrimetric analysis are:
A. $$Na_2Cr_2O_7$$
B. Oxalic acid
C. NaOH
D. $$FeSO_4 \cdot 6H_2O$$
E. Sodium tetraborate
Choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 57
The major product (P) in the following reaction is:
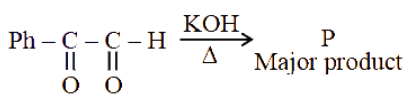
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 58
In the following series of reactions identify the major products A & B respectively.
Bromobenzene $$\xrightarrow{SO_3, H_2SO_4}$$ A (Major product) $$\xrightarrow{Br_2, Fe}$$ B (Major product)
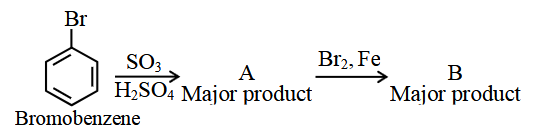
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 59
The standard cell potential ($$E^\ominus_{\text{cell}}$$) of a fuel cell based on the oxidation of methanol in air that has been used to power television relay station is measured as 1.21 V. The standard half cell reduction potential for $$O_2$$ ($$E^\circ_{O_2/H_2O}$$) is 1.229 V. Choose the correct statement:
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 60
Identify the diamagnetic octahedral complex ions from below:
A. $$[Mn(CN)_6]^{3-}$$
B. $$[Co(NH_3)_6]^{3+}$$
C. $$[Fe(CN)_6]^{4-}$$
D. $$[Co(H_2O)_3F_3]$$
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 61
In Dumas' method for estimation of nitrogen 0.4 g of an organic compound gave 60 mL of nitrogen collected at 300 K temperature and 715 mm Hg pressure. The percentage composition of nitrogen in the compound is (Given: Aqueous tension at 300 K = 15 mm Hg)
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 62
Mass of magnesium required to produce 220 mL of hydrogen gas at STP on reaction with excess of dil. HCl is (Given: Molar mass of Mg is 24 g mol$$^{-1}$$)
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 63
Given below are two statements:
Statement I: Wet cotton clothes made of cellulose based carbohydrate takes comparatively longer time to get dried than wet nylon polymer based clothes.
Statement II: Intermolecular hydrogen bonding with water molecule is more in nylon-based clothes than in the case of cotton clothes.
In the light of above statements, choose the $$Correct$$ answer from the options given below
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 64
Given below are two statements:
Statement I: $$CrO_3$$ is a stronger oxidizing agent than $$MoO_3$$.
Statement II: Cr(VI) is more stable than Mo(VI).
In the light of above statements, Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 65
Given below are two statements:
Statement I: Hyperconjugation is not a permanent effect.
Statement II: In general, greater the number of alkyl groups attached to a positively charged C-atom, greater is the hyperconjugation interaction and stabilization of the cation.
In the light of above statements, Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 66
Given below are two statements:
Statement I: When a system containing ice in equilibrium with water (liquid) is heated, heat is absorbed by the system and there is no change in the temperature until whole ice gets melted.
Statement II: At melting point of ice, there is absorption of heat in order to overcome intermolecular forces of attraction within the molecules of water in ice and kinetic energy of molecules is not increased at melting point.
In the light of above statements, Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 67
The sequence from the following that would result in giving predominantly 3,4,5-Tribromoaniline is:
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 68
The correct orders among the following are:
Atomic radius: $$B \lt Al \lt Ga \lt In \lt Tl$$
Electronegativity: $$Al \lt Ga \lt In \lt Tl \lt B$$
Density: $$Tl \lt In \lt Ga \lt Al \lt B$$
1st Ionisation Energy: $$In \lt Al \lt Ga \lt Tl \lt B$$
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 69
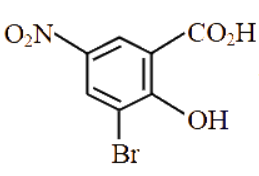
What is the correct IUPAC name of the following compound?
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 70
Consider the following statements related to temperature dependence of rate constants. Identify the correct statements:
A. The Arrhenius equation holds true only for an elementary homogenous reaction.
B. The unit of A is same as that of k in Arrhenius equation.
C. At a given temperature, a low activation energy means a fast reaction.
D. A and Ea as used in Arrhenius equation depend on temperature.
E. When $$E_a \gg RT$$, A and $$E_a$$ become interdependent.
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 71
X g of nitrobenzene on nitration gave 4.2 g of m-dinitrobenzene. X = ________ g. (nearest integer)
[Given: molar mass (in g mol$$^{-1}$$) C: 12, H: 1, O: 16, N: 14]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 72
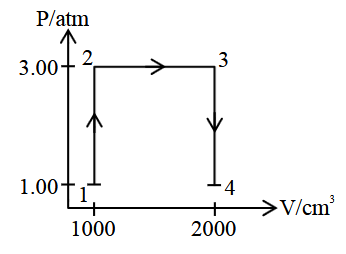
A perfect gas (0.1 mol) having $$\bar{C}_v = 1.50 R$$ (independent of temperature) undergoes the transformation from point 1 to point 4 as shown in the P-V diagram. If each step is reversible, the total work done (w) while going from point 1 to point 4 is $$(-) \text{____}$$ J (nearest integer).
[Given: $$R = 0.082$$ L atm $$K^{-1} mol^{-1}$$]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 73
A sample of n-octane (1.14 g) was completely burnt in excess of oxygen in a bomb calorimeter, whose heat capacity is 5 kJ $$K^{-1}$$. As a result of combustion reaction, the temperature of the calorimeter is increased by 5 K. The magnitude of the heat of combustion of octane at constant volume is ________ kJ mol$$^{-1}$$ (nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 74
Among Sc, Mn, Co and Cu, identify the element with highest enthalpy of atomisation. The spin only magnetic moment value of that element in its +2 oxidation state is ________ BM (in nearest integer).
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 2 - Chemistry - Question 75
The total number of structural isomers possible for the substituted benzene derivatives with the molecular formula $$C_9H_{12}$$ is ________.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




