NTA JEE Main 2025 April 3rd Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 51
Which of the following postulate of Bohr's model of hydrogen atom is not in agreement with quantum mechanical model of an atom ?
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 52
Given below are two statements :
Statement I : The N-N single bond is weaker and longer than that of P-P single bond
Statement II : Compounds of group 15 elements in +3 oxidation states readily undergo disproportionation reactions.
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 53
Given below are two statements
Statement I : A catalyst cannot alter the equilibrium constant $$(K_c)$$ of the reaction, temperature remaining constant
Statement II : A homogeneous catalyst can change the equilibrium composition of a system temperature remaining constant
In the light of the above statements, choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 54
The metal ions that have the calculated spin only magnetic moment value of 4.9 B.M. are
A. $$Cr^{2+}$$
B. $$Fe^{2+}$$
C. $$Fe^{3+}$$
D. $$Co^{2+}$$
E. $$Mn^{3+}$$
Choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 55
In a reaction $$A + B \to C$$, initial concentrations of A and B are related as $$[A]_0 = 8[B]_0$$. The half lives of A and B are 10 min and 40 min respectively. If they start to disappear at the same time, both following first order kinetics, after how much time will the concentration of both the reactants be same?
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 56
Which of the following is the correct structure of L-fructose ?
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 57
Identify the correct statements from the following
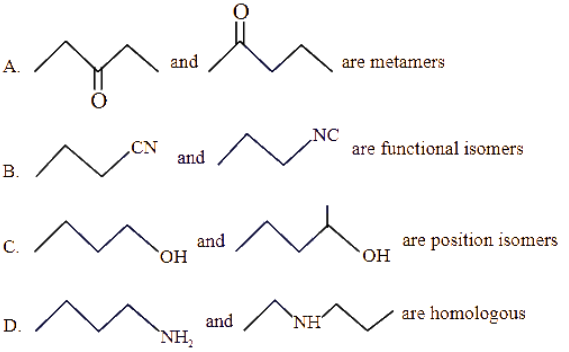
Choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 58
Among $$10^{-9}$$ g (each) of the following elements, which one will have the highest number of atoms?
Element : Pb, Po, Pr and Pt
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 59
Which of the following statements are correct?
A. The process of the addition of an electron to a neutral gaseous atom is always exothermic
B. The process of removing an electron from an isolated gaseous atom is always endothermic
C. The 1st ionization energy of the boron is less than that of the beryllium
D. The electronegativity of C is 2.5 in $$CH_4$$ and $$CCl_4$$
E. Li is the most electropositive among elements of group I
Choose the correct answer from the options given below
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 60
Which of the following properties will change when system containing solution 1 will become solution 2 ?
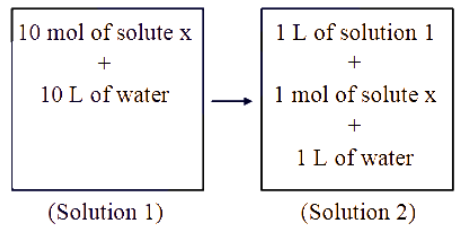
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 61
Number of molecules from below which cannot give iodoform reaction is :
Ethanol, Isopropyl alcohol, Bromoacetone, 2-Butanol, 2-Butanone, Butanal, 2-Pentanone, 3-Pentanone, Pentanal and 3-Pentanol
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 62
Identify [A], [B], and [C], respectively in the following reaction sequence :
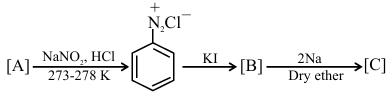
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 63
In the following reactions, which one is NOT correct?
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 64
The correct order of the complexes $$[Co(NH_3)_4(H_2O)]^{3+}$$ (A), $$[Co(NH_3)_6]^{3+}$$ (B), $$[Co(CN)_6]^{3-}$$ (C) and $$[CoCl(NH_3)_5]^{2+}$$ (D) in terms of wavelength of light absorbed is :
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 65
2 moles each of ethylene glycol and glucose are dissolved in 500 g of water. The boiling point of the resulting solution is :
(Given : Ebullioscopic constant of water = 0.52 K kg mol$$^{-1}$$)
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 66
Which compound would give 3-methyl-6-oxoheptanal upon ozonolysis ?
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 67
Match the LIST-I with LIST-II.
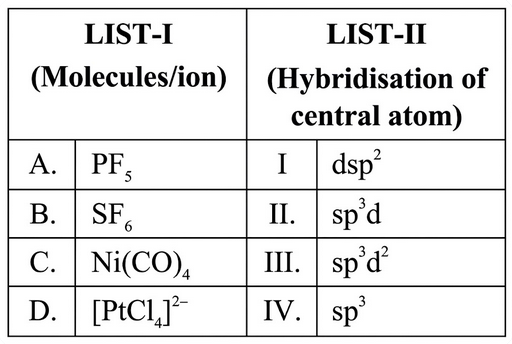
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 68
The least acidic compound, among the following is:
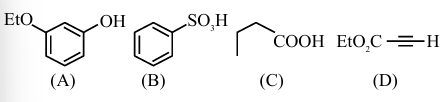
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 69
Correct order of limiting molar conductivity for cations in water at 298 K is :
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 70
During estimation of nitrogen by Dumas' method of compound X (0.42 g) :
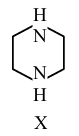
_________ mL of $$N_2$$ gas will be liberated at STP. (nearest integer)
(Given molar mass in g mol$$^{-1}$$ : C : 12, H : 1, N : 14)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 71
0.5 g of an organic compound on combustion gave 1.46 g of $$CO_2$$ and 0.9 g of $$H_2O$$. The percentage of carbon in the compound is _________. (Nearest integer)
[Given : Molar mass (in g mol$$^{-1}$$) C : 12, H : 1, O : 16]
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 72
The number of optical isomers exhibited by the iron complex (A) obtained from the following reaction is _________.
$$FeCl_3 + KOH + H_2C_2O_4 \to A$$
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 73
Given :
$$\Delta H^{\ominus}_{\text{sub}}[C(\text{graphite})] = 710$$ kJ mol$$^{-1}$$
$$\Delta_{C-H} H^{\ominus} = 414$$ kJ mol$$^{-1}$$
$$\Delta_{H-H} H^{\ominus} = 436$$ kJ mol$$^{-1}$$
$$\Delta_{C=C} H^{\ominus} = 611$$ kJ mol$$^{-1}$$
The $$\Delta H_f^{\ominus}$$ for $$CH_2=CH_2$$ is _________ kJ mol$$^{-1}$$
(nearest integer value)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 74
Consider the following reactions
$$A + NaCl + H_2SO_4 \to CrO_2Cl_2 + \text{Side Products}$$ (Little amount)
$$CrO_2Cl_{2(Vapour)} + NaOH \to B + NaCl + H_2O$$
$$B + H^+ \to C + H_2O$$
The number of terminal 'O' present in the compound 'C' is _________.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 3rd Shift 1 - Chemistry - Question 75
In the following system,
$$PCl_5(g) \rightleftharpoons PCl_3(g) + Cl_2(g)$$ at equilibrium, upon addition of xenon gas at constant T & p, the concentration of
.webp)
.webp)
.webp)
.webp)




