NTA JEE Main 2025 April 02 Shift 2 - Chemistry
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 51
When a concentrated solution of sulphanilic acid and 1-naphthylamine is treated with nitrous acid (273 K) and acidified with acetic acid, the mass (g) of 0.1 mole of product formed is :
(Given molar mass in g mol$$^{-1}$$ H : 1, C : 12, N : 14, O : 16, S : 32)
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 52
The d-orbital electronic configuration of the complex among $$[Co(en)_3]^{3+}$$, $$[CoF_6]^{3-}$$, $$[Mn(H_2O)_6]^{2+}$$ and $$[Zn(H_2O)_6]^{2+}$$ that has the highest CFSE is :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 53
Given below are two statements :
Statement (I) : Neopentane forms only one monosubstituted derivative.
Statement (II) : Melting point of neopentane is higher than n-pentane.
In the light of the above statements, choose the most appropriate answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 54
Which among the following molecules is (a) involved in $$sp^3d$$ hybridization, (b) has different bond lengths and (c) has lone pair of electrons on the central atom ?
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 55
Formation of $$Na_4[Fe(CN)_5NOS]$$, a purple coloured complex formed by addition of sodium nitroprusside in sodium carbonate extract of salt indicates the presence of :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 56
In 3,3-dimethylhex-1-ene-4-yne, there are _____ $$sp^3$$, _____ $$sp^2$$ and _____ $$sp$$ hybridised carbon atoms respectively :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 57
Which of the following statements are true ?
(A) The subsidiary quantum number $$l$$ describes the shape of the orbital occupied by the electron.
(B)
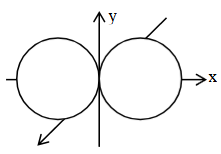
is the boundary diagram of the $$2p_x$$ orbital.
(C) The + and - signs in the wave function of the $$2p_x$$ orbital refer to charge.
(D) The wave function of $$2p_x$$ orbital is zero everywhere in the xy plane.
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 58
The type of hybridization and the magnetic property of $$[MnCl_6]^{3-}$$ are :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 59
Consider the following reactions. From these reactions which reaction will give carboxylic acid as a major product ?
(A) $$R-C \equiv N \xrightarrow[\text{mild condition}]{(i) H^+/H_2O}$$
(B) $$R-MgX \xrightarrow[(ii) H_3O^+]{(i) CO_2}$$
(C) $$R-C \equiv N \xrightarrow[(ii) H_3O^+]{(i) SnCl_2/HCl}$$
(D) $$R-CH_2-OH \xrightarrow{PCC}$$
(E)
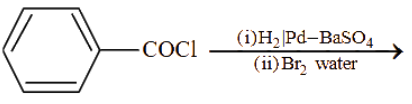
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 60
Electronic configuration of four elements A, B, C and D are given below :
(A) $$1s^2 2s^2 2p^3$$
(B) $$1s^2 2s^2 2p^4$$
(C) $$1s^2 2s^2 2p^5$$
(D) $$1s^2 2s^2 2p^2$$
Which of the following is the correct order of increasing electronegativity (Pauling's scale) ?
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 61
Match List-I with List-II.
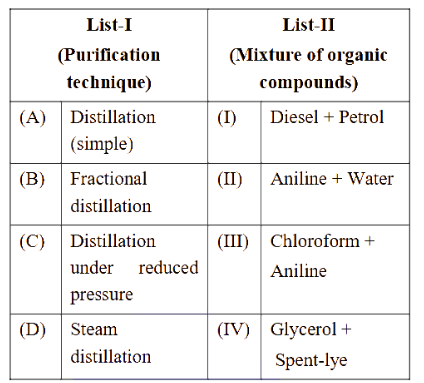
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 62
'x' g of NaCl is added to water in a beaker with a lid. The temperature of the system is raised from 1°C to 25°C. Which of the following plots, is best suited for the change in the molarity (M) of the solution with respect to temperature ?
[Consider the solubility of NaCl remains unchanged over the temperature range]
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 63
Arrange the following in order of magnitude of work done by the system / on the system at constant temperature :
(a) $$|w_{\text{reversible}}|$$ for expansion in infinite stage.
(b) $$|w_{\text{irreversible}}|$$ for expansion in single stage.
(c) $$|w_{\text{reversible}}|$$ for compression in infinite stage.
(d) $$|w_{\text{irreversible}}|$$ for compression in single stage.
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 64
Reactant A converts to product D through the given mechanism (with the net evolution of heat) :
$$A \to B$$ slow ; $$\Delta H = +ve$$
$$B \to C$$ fast ; $$\Delta H = -ve$$
$$C \to D$$ fast ; $$\Delta H = -ve$$
Which of the following represents the above reaction mechanism ?
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 65
The nature of oxide $$(TeO_2)$$ and hydride $$(TeH_2)$$ formed by Te, respectively are :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 66
Match List-I with List-II.
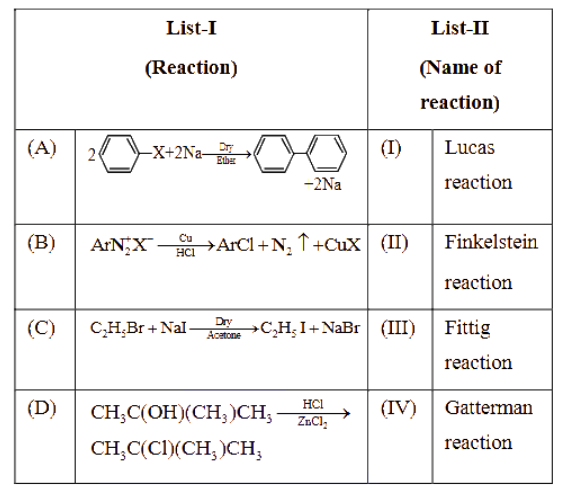
Choose the correct answer from the options given below :
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 67
Consider the following chemical equilibrium of the gas phase reaction at a constant temperature :
$$A(g) \rightleftharpoons B(g) + C(g)$$
If p being the total pressure, $$K_p$$ is the pressure equilibrium constant and $$\alpha$$ is the degree of dissociation, then which of the following is true at equilibrium ?
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 68
Which of the following graphs correctly represents the variation of thermodynamic properties of Haber's process ?
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 69
A tetrapeptide "x" on complete hydrolysis produced glycine (Gly), alanine (Ala), valine (Val), leucine (Leu) in equimolar proportion each. The number of tetrapeptides (sequences) possible involving each of these amino acids is
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 70
In Dumas' method for estimation of nitrogen, 0.5 gram of an organic compound gave 60 mL of nitrogen collected at 300 K temperature and 715 mm Hg pressure. The percentage composition of nitrogen in the compound (Aqueous tension at 300 K = 15 mm Hg) is
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 71
For the reaction $$A \to B$$ the following graph was obtained. The time required (in seconds) for the concentration of A to reduce to 2.5 g L$$^{-1}$$ (if the initial concentration of A was 50 g L$$^{-1}$$) is _________.
(Nearest integer)
Given : $$\log 2 = 0.3010$$
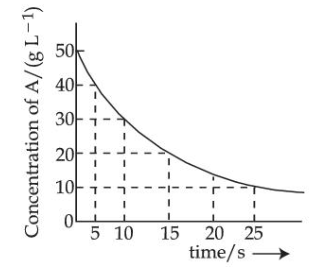
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 72
0.2 % (w/v) solution of NaOH is measured to have resistivity 870.0 m$$\Omega$$ m. The molar conductivity of the solution will be _________ $$\times 10^2$$ mS dm$$^2$$ mol$$^{-1}$$.
(Nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 73
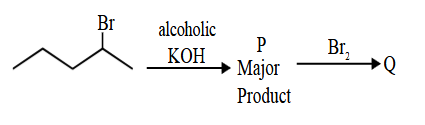
Consider the above sequence of reactions. 151 g of 2-bromopentane is made to react. Yield of major product P is 80% whereas Q is 100%.
Mass of product Q obtained is _________ g.
(Given molar mass in g mol$$^{-1}$$ H: 1, C: 12, O: 16, Br: 80)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 74
When 1 g each of compounds AB and $$AB_2$$ are dissolved in 15 g of water separately, they increased the boiling point of water by 2.7 K and 1.5 K respectively. The atomic mass of A (in amu) is _________ $$\times 10^{-1}$$.
(Nearest integer)
(Given : Molal boiling point elevation constant is 0.5 K kg mol$$^{-1}$$)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 2 - Chemistry - Question 75
The spin-only magnetic moment value of $$M^{n+}$$ ion formed among Ni, Zn, Mn and Cu that has the least enthalpy of atomisation is _________. (in nearest integer)
Here n is equal to the number of diamagnetic complexes among $$K_2[NiCl_4]$$, $$[Zn(H_2O)_6]Cl_2$$, $$K_3[Mn(CN)_6]$$ and $$[Cu(PPh_3)_3I]$$
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




