NTA JEE Main 2025 April 02 Shift 1 - Chemistry
For the following questions answer them individually
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 51
Designate whether each of the following compounds is aromatic or not aromatic.
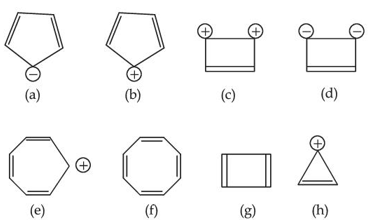
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 52
An optically active alkyl halide $$C_4H_9Br$$ [A] reacts with hot KOH dissolved in ethanol and forms alkene [B] as major product which reacts with bromine to give dibromide [C]. The compound [C] is converted into a gas [D] upon reacting with alcoholic NaNH$$_2$$. During hydration 18 gram of water is added to 1 mole of gas [D] on warming with mercuric sulphate and dilute acid at 333 K to form compound [E]. The IUPAC name of compound [E] is:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 53
The property/properties that show irregularity in first four elements of group-17 is/are:
(A) Covalent radius
(B) Electron affinity
(C) Ionic radius
(D) First ionization energy
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 54
Which of the following graph correctly represents the plots of $$K_H$$ at 1 bar gases in water versus temperature?
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 55
According to Bohr's model of hydrogen atom, which of the following statement is incorrect?
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 56
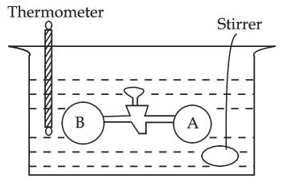
Two vessels A and B are connected via stopcock. The vessel A is filled with a gas at a certain pressure. The entire assembly is immersed in water and is allowed to come to thermal equilibrium with water. After opening the stopcock the gas from vessel A expands into vessel B and no change in temperature is observed in the thermometer. Which of the following statement is true?
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 57
A solution is made by mixing one mole of volatile liquid A with 3 moles of volatile liquid B. The vapour pressure of pure A is 200 mm Hg and that of the solution is 500 mm Hg. The vapour pressure of pure B and the least volatile component of the solution, respectively, are:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 58
$$CaCO_3(s) + 2HCl(aq) \rightarrow CaCl_2(aq) + CO_2(g) + H_2O(l)$$
Consider the above reaction, what mass of $$CaCl_2$$ will be formed if 250 mL of 0.76 M HCl reacts with 1000 g of $$CaCO_3$$?
(Given: Molar mass of Ca, C, O, H and Cl are 40, 12, 16, 1 and 35.5 g mol$$^{-1}$$, respectively)
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 59
If equal volumes of $$AB_2$$ and XY (both are salts) aqueous solutions are mixed, which of the following combination will give a precipitate of $$AY_2$$ at 300 K?
(Given $$K_{sp}$$ (at 300 K) for $$AY_2 = 5.2 \times 10^{-7}$$)
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 60
Among $$SO_2$$, $$NF_3$$, $$NH_3$$, $$XeF_2$$, $$ClF_3$$ and $$SF_4$$, the hybridization of the molecule with non-zero dipole moment and highest number of lone pairs of electrons on the central atom is:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 61
Given below are two statements:
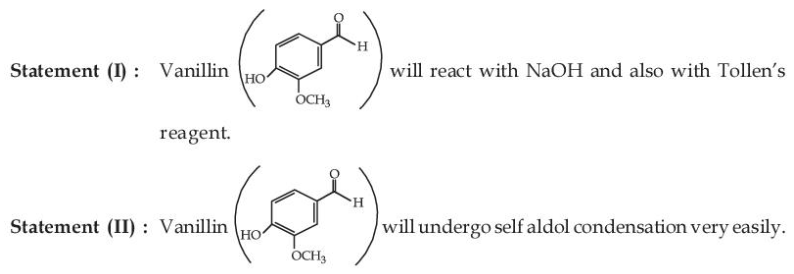
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 62
Identify the correct statement among the following:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 63
The correct order of basic nature on aqueous solution for the bases $$NH_3$$, $$H_2N-NH_2$$, $$CH_3CH_2NH_2$$, $$(CH_3CH_2)_2NH$$ and $$(CH_3CH_2)_3N$$ is:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 64
Given below are two statements:
Statement (I): The metallic radius of Al is less than that of Ga.
Statement (II): The ionic radius of Al$$^{3+}$$ is less than that of Ga$$^{3+}$$.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 65
Given below are two statements:
Statement (I): In octahedral complexes, when $$\Delta_o \lt P$$ high spin complexes are formed. When $$\Delta_o \gt P$$ low spin complexes are formed.
Statement (II): In tetrahedral complexes because of $$\Delta_t \lt P$$, low spin complexes are rarely formed.
In the light of the above statements, choose the most appropriate answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 66
Choose the correct tests with respective observations.
(A) $$CuSO_4$$ (acidified with acetic acid) + $$K_4[Fe(CN)_6]$$ $$\rightarrow$$ Chocolate brown precipitate.
(B) $$FeCl_3 + K_4[Fe(CN)_6]$$ $$\rightarrow$$ Prussian blue precipitate.
(C) $$ZnCl_2 + K_4[Fe(CN)_6]$$, neutralised with $$NH_4OH$$ $$\rightarrow$$ White or bluish white precipitate.
(D) $$MgCl_2 + K_4[Fe(CN)_6]$$ $$\rightarrow$$ Blue precipitate.
(E) $$BaCl_2 + K_4[Fe(CN)_6]$$, neutralised with NaOH $$\rightarrow$$ White precipitate.
Choose the correct answer from the options given below:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 67
On complete combustion 1.0 g of an organic compound (X) gave 1.46 g of $$CO_2$$ and 0.567 g of $$H_2O$$. The empirical formula mass of compound (X) is ________ g.
(Given molar mass in g mol$$^{-1}$$ C: 12, H: 1, O: 16)
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 68
Consider the following compound (X):
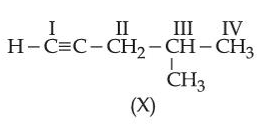
The most stable and least stable carbon radicals, respectively, produced by homolytic cleavage of corresponding C-H bond are:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 69
Consider the following molecules:
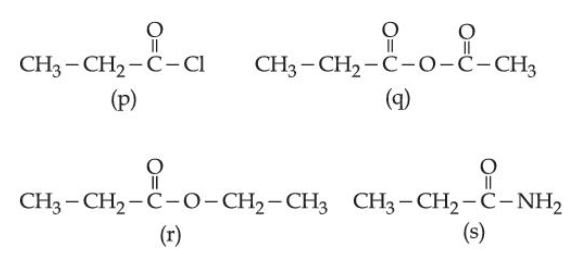
The correct order of rate of hydrolysis is:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 70
A molecule with the formula $$AX_4Y$$ has all its elements from p-block. Element $$A$$ is rarest, monoatomic, non-radioactive from its group and has the lowest ionization enthalpy value among$$ A, X$$ and $$Y$$. Elements $$X$$ and $$Y$$ have first and second highest electronegativity values respectively among all the known elements. The shape of the molecule is:
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 71
A transition metal $$(M)$$ among $$Mn, Cr, Co$$ and $$Fe$$ has the highest standard electrode potential ($$M^{3+}/M^{2+}$$). It forms a metal complex of the type $$[M(CN)_6]^{4-}$$. The number of electrons present in the $$e_g$$ orbital of the complex is ______.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 72
Consider the following electrochemical cell at standard condition.
$$Au(s)|QH_2,Q|NH_4X(0.01M)||Ag^+(1M)|Ag(s)$$
$$E_{cell} = +0.4V$$
The couple $$QH_2/Q$$ represents quinhydrone electrode, the half cell reaction is given below:
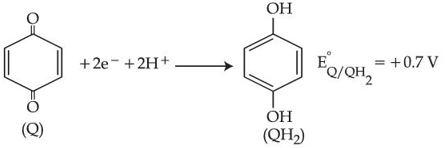
[Given: $$E^o_{Ag^+/Ag} = +0.8V$$ and $$\frac{2.303RT}{F} = 0.06V$$]
The $$pK_b$$ value of the ammonium halide salt ($$NH_4X$$) used here is ______. (nearest integer)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 73
0.1 mol of the following given antiviral compound (P) will weigh _______ $$\times 10^{-1}$$ g.
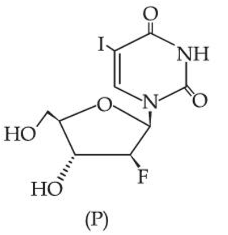
(Given: molar mass in g mol$$^{-1}$$ H: 1, C: 12, N: 14, O: 16, F: 19, I: 127)
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 74
Consider the following equilibrium,
$$CO(g) + 2H_2(g) \rightleftharpoons CH_3OH(g)$$
0.1 mol of CO along with a catalyst is present in a 2 dm$$^3$$ flask maintained at 500 K. Hydrogen is introduced into the flask until the pressure is 5 bar and 0.04 mol of $$CH_3OH$$ is formed. The $$K_p^0$$ is _______ $$\times 10^{-3}$$. (nearest integer)
Given $$R = 0.08$$ dm$$^3$$ bar K$$^{-1}$$ mol$$^{-1}$$
Assume only methanol is formed as the product and the system follows ideal gas behaviour.
789
456
123
0.-
Clear All
NTA JEE Main 2025 April 02 Shift 1 - Chemistry - Question 75
For the reaction $$A \rightarrow$$ products.
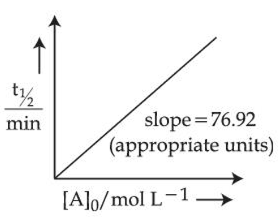
The concentration of A at 10 minutes is _______ $$\times 10^{-3}$$ mol L$$^{-1}$$. (nearest integer).
The reaction was started with 2.5 mol L$$^{-1}$$ of A.
789
456
123
0.-
Clear All
.webp)
.webp)
.webp)
.webp)




